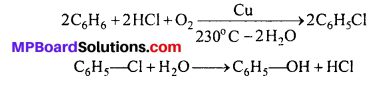
MP Board Class 11th Chemistry Solutions Chapter 14 पर्यावरणीय रसायन
पर्यावरणीय रसायन NCERT अभ्यास प्रश्न
प्रश्न 1.
पर्यावरणीय रसायन की परिभाषा दीजिए।
उत्तर:
पर्यावरणीय रसायन विज्ञान की वह शाखा है जो पर्यावरण में रासायनिक परिवर्तनों से सम्बन्धित होती हैं। इसमें हमारा परिवेश सम्मिलित होता है, जैसे कि वायु, जल, मृदा, जंगल, सूर्य का प्रकाश आदि।
प्रश्न 2.
क्षोभमण्डलीय प्रदूषण की 100 शब्दों में व्याख्या कीजिए।
उत्तर:
क्षोभमण्डलीय प्रदूषण वायु में उपस्थित अवांछनीय ठोस अथवा गैस के कणों के कारण उत्पन्न होता है। क्षोभमण्डल में मुख्यतः निम्नलिखित गैसीय तथा कणिकीय प्रदूषण उपस्थित होते हैं
- गैसीय वायुप्रदूषक – ये सल्फर, नाइट्रोजन तथा कार्बन डाईऑक्साइड, हाइड्रोजन सल्फाइड, हाइड्रोकार्बन, ओजोन तथा अन्य ऑक्सीकारक होते हैं।
- कणिकीय प्रदूषक – ये धूल, धूम, कोहरा, फुहारा, धुआँ आदि होते हैं।
![]()
प्रश्न 3.
कार्बन मोनोऑक्साइड, कार्बन डाइऑक्साइड की अपेक्षा अधिक खतरनाक होती है। क्यों?
उत्तर:
कार्बन मोनोऑक्साइड एक विषैली गैस है। यह रक्त में उपस्थित हीमोग्लोबिन के साथ संयुक्त होकर कार्बोक्सीहीमोग्लोबीन (COHb) बनाती है। यह ऑक्सीहीमोग्लोबिन की अपेक्षा 300 गुना अधिक स्थायी है। जब रक्त में कार्बोक्सीहीमोग्लोबिन की मात्रा 3-4% तक हो जाती है तब रक्त में ऑक्सीजन ले जाने की क्षमता अत्यधिक कम हो जाती है। ऑक्सीजन की इस न्यूनता से सिरदर्द, नेत्रदृष्टि की क्षीणता, तंत्रकीय आवेग में न्यूनता, हृदयवाहिका में तंत्र अवस्था आदि रोग उत्पन्न हो जाते हैं। कार्बन डाइऑक्साइड गैस हीमोग्लोबिन के साथ संयुक्त नहीं होती है। अतः यह एक कम हानिकारक प्रदूषक है। वातावरण में इसकी मात्रा में वृद्धि के कारण मुख्यतः वैश्विक गर्मी (भूमंडलीय ताप वृद्धि) हो रही है।
प्रश्न 4.
ग्रीन हाऊस प्रभाव के लिए कौन-सी गैसें उत्तरदायी हैं ? सूचीबद्ध कीजिए।
उत्तर:
निम्नलिखित गैसों द्वारा हरित गृह प्रभाव उत्पन्न किया जा सकता है, जो कि ऊष्मा ऊर्जा को रोकने में समर्थ होती है –
- कार्बन डाइऑक्साइड
- मेथेन
- ओजोन
- क्लोरोफ्लुओरोकार्बन के यौगिक (CFCs)
- जल वाष्प।
प्रश्न 5.
अम्लवर्षा मूर्तियों तथा स्मारकों को कैसे दुष्प्रभावित करती है?
उत्तर:
भारत में मूर्तियाँ तथा स्मारक संगमरमर अर्थात् CaCO3 के बने होते हैं। जिसके परितः वायु में मुख्यतः सल्फर तथा नाइट्रोजन के ऑक्साइडों की अधिक मात्रा होती है। इसका मुख्य कारण इन क्षेत्रों के आसपास औद्योगिक इकाइयों तथा ऊर्जा संयंत्रों की मात्रा अधिक होना है। सल्फर तथा नाइट्रोजन के ऑक्साइड अम्लीय होते हैं। SO2 तथा NO2 जल के साथ ऑक्सीकरण क्रिया करके खनिज अम्ल बनाते हैं जो अम्ल वर्षा के मुख्य स्त्रोत हैं।
2SO2(g) + O2(g) + 2H2O(l) → 2H2SO4(aq)
4NO2(g) + O2(g) + 2H2O → 4HNO3(aq)
यह अम्ल वर्षा, मूर्तियों तथा स्मारकों के संगमरमर के साथ क्रिया करके उन्हें नष्ट करती हैं।
CaCO3 + H2SO4 → CaSO4 + H3O+ CO2
प्रश्न 6.
धूम-कोहरा क्या है? सामान्य धूम-कोहरा, प्रकाश रासायनिक धूम-कोहरे से कैसे भिन्न है?
उत्तर:
धूम-कोहरा शब्द धूम तथा कोहरा से उत्पन्न हुआ है। यह विश्व के अनेक शहरों में पाया जाने वाला वायु प्रदूषक है। धूम-कोहरा निम्न दो प्रकार का होता है –
1. सामान्य धूम-कोहरा:
यह ठंडे आर्द्र वातावरण में उत्पन्न होता है। यह धुएँ, कोहरे तथा So, का मिश्रण है। रासायनिक रूप से यह अपचायक धूम-कोहरा है।
2. प्रकाश रासायनिक धूम-कोहरा:
यह गर्म, शुष्क तथा सूर्य की पर्याप्त रोशनी वाले वातावरण में उत्पन्न होता है। इसके मुख्य अवयव ओजोन, फॉर्मेल्डिहाइड, एक्रोलीन तथा परॉक्सीऐसीटिल नाइट्रेट (PAN) (असंतृप्त हाइड्रोकार्बनों पर सूर्य के प्रकाश की क्रिया से उत्पन्न) तथा नाइट्रोजन ऑक्साइड (वाहनों तथा कारखानों से उत्पन्न) होते हैं। रासायनिक रूप से यह ऑक्सीकारक धूम-कोहरा है क्योंकि इसमें ऑक्सीकारक अभिकर्मकों की सान्द्रता उच्च रहती है।
![]()
प्रश्न 7.
प्रकाश रासायनिक धूम-कोहरे के निर्माण के दौरान होने वाली अभिक्रिया लिखिए।
उत्तर:
जब जीवाश्म ईंधनों का दहन होता है, तब पृथ्वी के वातावरण में कई प्रदूषक उत्सर्जित होते हैं। इनमें से दो प्रदूषक हाइड्रोकार्बन एवं नाइट्रिक ऑक्साइड (NO) है। जब इन प्रदूषकों का स्तर पर्याप्त ऊँचा हो जाता है, तब सूर्य प्रकाश से इसकी अन्योन्य क्रिया के कारण श्रृंखला अभिक्रिया होती है, जिसमें NO2 नाइट्रोजन डाइऑक्साइड NO2 में परिवर्तित हो जाती है। यह NO2 सूर्य प्रकाश से ऊर्जा ग्रहण कर पुनः नाइट्रिक ऑक्साइड एवं मुक्त ऑक्सीजन में विघटित हो जाती है।
NO2(g) \(\underrightarrow { hv } \) NO(g) + O(g) …(i)
ऑक्सीजन परमाणु अत्यधिक क्रियाशील होने के कारण O, के साथ संयुक्त होकर ओजोन में परिवर्तित हो जाता है।
O(g) + O2(g) → O3(g) ….(ii)
निर्मित O3, शीघ्रतापूर्वक अभिक्रिया (i) में विरचित NO(g) के साथ अभिक्रिया कर पुन: NO2 बनाती है। NO2 एक भूरी गैस है, जिसका उच्च स्तर धुंध का कारण हो सकता है।
NO(g) + O3(g) → NO2(g) + O3(g)
ओजोन एक जहरीली गैस है। NO, एवं 0, दोनों ही प्रबल ऑक्सीकारक हैं। जैसे –

प्रश्न 8.
प्रकाश रासायनिक धूम-कोहरे के दुष्परिणाम क्या है? इन्हें कैसे नियन्त्रित किया जा सकता है?
उत्तर:
प्रकाश रासायनिक धूम-कोहरे के दुष्परिणाम-प्रकाश रासायनिक धूम-कोहरा के प्रमुख घटक ओजोन, नाइट्रोजन के ऑक्साइड, एक्रोलीन, फॉर्मेल्डिहाइड और परॉक्सीऐसीटिल नाइट्रेट (PAN) आदि हैं। ये हानिकारक प्रभावों के लिए जिम्मेदार होते हैं। इनमें से कुछ निम्न हैं –
- ओजोन और नाइट्रिक ऑक्साइड नाक एवं गले में उत्तेजना पैदा करते हैं। इनके उच्च सान्द्रण से सिर और सीने में दर्द होने लगता है।
- प्रकाश रासायनिक धूम-कोहरा के संघटक गैसें सामान्यतः गले में शुष्कता पैदा करती है तथा ये श्वसन समस्याओं के लिए जिम्मेदार होती हैं।
- प्रकाश रासायनिक धूम-कोहरा पादप जीवन को बहुत क्षति पहुँचाती है।
- ये धातुओं, इमारती पदार्थों, रबर और रंजित सतह आदि के संक्षारण के लिए भी जिम्मेवार होती हैं।
प्रकाश रासायनिक धूम-कोहरा नियन्त्रित करने के उपाय:
निम्नलिखित उपायों द्वारा प्रकाश रासायनिक धूम-कोहरा से उत्पन्न प्रदूषण को कुछ मात्रा तक रोका जा सकता है –
- वाहनों के इंजनों में उत्प्रेरकीय परिवर्तकों के उपयोग से NO2 और कुछ हाइड्रोकार्बन जैसे प्राथमिक पूर्वगामी नियन्त्रित हो जाएंगे। इससे PAN जैसे द्वितीयक पूर्वगामी का विरचन स्वतः नियन्त्रित हो जाएगा।
- कुछ पादप जैसे पाईनस, पायरस, विटिस, क्वेरकस आदि काफी खतरनाक नाइट्रोजन के ऑक्साइडों का उपापचय करने में समर्थ होते हैं। इनका रोपण निश्चित रूप से वायुमण्डल में इन गैसों को फैलने से रोकने में सहायक होगा।
प्रश्न 9.
क्षोभमण्डल पर ओजोन-परत के क्षय में होने वाली अभिक्रिया कौन-सी है?
उत्तर:
समतापमण्डल में उपस्थित फ्रीऑन जैसे क्लोरोफ्लुओरोकार्बन ओजोन के साथ रासायनिक अभिक्रिया करते हैं। ये मुक्त मूलक प्रकृति के होते हैं तथा अभिक्रिया पराबैंगनी विकिरणों की उपस्थिति में होती है।
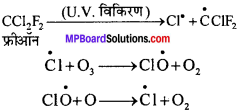
चूँकि ओजोन रासायनिक अभिक्रिया में भाग लेती है, इसलिए ओजोन परत का क्रमिक अवक्षय होता है।
प्रश्न 10.
ओजोन छिद्र से आप क्या समझते हैं? इसके परिणाम क्या हैं?
उत्तर:
ओजोन छिद्र हानिकारक पराबैंगनी विकिरणों द्वारा ओजोन परत के ध्वंश को सूचित करता है। इससे हमारे चारों ओर स्थित ओजोन की परत में वस्तुतः छिद्र हो जाएंगे। इसके परिणामस्वरूप, हानिकारक विकिरणों से त्वचा कैंसर, दृष्टिदोष आदि उत्पन्न होंगे और यह हमारे प्रतिरक्षा तंत्र (Immune system) को प्रभावित करेगा।
ओजोन पर्त के दुष्प्रभाव:
- पराबैंगनी किरणों का बिना रोकथाम के सीधे धरती पर प्रवेश होने पर विभिन्न रोगों का कारण हो सकता है। जैसे-कैंसर।
- पराबैंगनी किरणें शरीर की प्रतिरोधक क्षमता को कम कर देती हैं।
- C.EC. ग्रीन हाऊस प्रभाव को कम करती है, परिणामस्वरूप धरती के तापमान में वृद्धि होती है।
नियत्रंण:
- C.E.C. का उत्पादन कम करना।
- C.F.C. का विकल्प ढूँढ़ना।
प्रश्न 11.
क्या आपने अपने क्षेत्र में जल-प्रदूषण देखा है, इसे नियन्त्रित करने के कौन-कौन से उपाय हैं?
उत्तर:
पेपर, कपड़ा तथा रासायनिक उद्योगों से अनेक अपशिष्ट पदार्थ जल में मिलकर इसको प्रदूषित करते हैं। अत: जल प्रदूषण की रोकथाम के लिए उद्योग अपशिष्ट को नदी नालों में न डालकर इनका समुचित विस्थापन करना चाहिए। कपड़े धोने के जैव निम्नीकरण साबुन व अपमार्जक का प्रयोग करना चाहिए न कि जैव अनिम्नीकरण पदार्थों का। जल का pH मान ज्ञात करना चाहिए। शुद्ध जल का pH मान 7 है यदि जल का pH मान 7 से कम है तब इसमें उपस्थित अशुद्धियाँ अम्लीय है जैसे SO2, H2S आदि।
यदि जल का pH मान 7 से अधिक है, तब अशुद्धियों की प्रकृति क्षारीय है जैसे साबुन व अपमार्जक। ऐसी स्थिति में प्रदूषण नियंत्रक बोर्ड को सूचित कर रोकथाम करना चाहिए। रासायनिक उर्वरकों की बजाय देशी खाद का उपयोग करना चाहिए। DDT, मैलाथीआन आदि का उपयोग नहीं करना चाहिए। KMnO4 या विरंचक चूर्ण से जल का शुद्धिकरण करना चाहिए।
प्रश्न 12.
आप ‘जैव रासायनिक ऑक्सीजन आवश्यकता’ (B.O.D.) से क्या समझते हैं?
उत्तर:
इसे निम्न प्रकार से परिभाषित किया जा सकता है –
- यह जल में घुलित ऑक्सीजन की मिलीग्राम में वह मात्रा है, जो एक लीटर जल में उपस्थित कार्बनिक पदार्थ के 5 दिन तक 20°C ताप विघटन के लिए आवश्यक होती है।
- शुद्ध जल का B.O.D. 3 ppm तक होता है। यदि स्तर अधिक हो तो यह जल में कार्बनिक अपशिष्ट की उपस्थिति का सूचक है।
![]()
प्रश्न 13.
क्या आपने आस-पास के क्षेत्र में भूमि-प्रदूषण देखा है ? आप भूमि-प्रदूषण को नियन्त्रित करने के लिए क्या प्रयास करेंगे?
उत्तर:
हाँ, इसको निम्नलिखित विधियों द्वारा नियंत्रित किया जा सकता है –
1. कीटनाशी तथा पीड़कनाशी जिनका प्रयोग फसलों की रक्षा के लिए किया जाता है भूमि प्रदूषण फैलाते हैं। शाकनाशी (दुर्बलनाशी) भी भूमि प्रदूषण फैलाते हैं। अतः इसके समुचित प्रयोग की आवश्यकता है।
2. द्वितीय विश्व युद्ध के पश्चात् डी.डी.टी. का प्रयोग कृषि में कीट, सेडेंट, खरपतवार तथा फसलों के अनेक रोगों को नियंत्रण के रूप में किया जाने लगा। इसके प्रतिकूल प्रभावों के कारण भारत में इसका प्रयोग प्रतिबंधित हो गया है। एल्ड्रीन तथा डाइएल्ड्रीन जैसे पीड़कनाशी वास्तव में जैव विष हैं। ये जल में अविलेय तथा अजैवनिम्नीकरण होते हैं। इनके कारण जीवों में उपापचयी तथा शरीर क्रियात्मक अवस्थाएँ उत्पन्न हो जाती हैं। आजकल ऑर्गेनो फॉस्फेट्स तथा कार्बोनेट्स को पीड़कनाशी के रूप में प्रयोग किया जा रहा है। ये अधिक जैवनिम्नीकरण वाले यौगिक हैं परन्तु ये गंभीर स्नायु जीव विष हैं अतः ये मानव के लिए हानिकारक हैं। अतः उर्वरक, डिटर्जेंट पीड़कनाशी, बहुलक आदि जैसे रसायनों का प्रयोग केवल तभी किया जाना चाहिए जबकि उनकी अति आवश्यकता हो।
3. जैवनिम्नीकृत घरेलू अवशिष्ट को जमीन के गड्ढों में दबाना चाहिए।
4. अजैवनिम्नीकृत अपशिष्टों का पुर्नचक्रण किया जाना चाहिए।
5. पॉलीथीन के प्रयोग से बचना चाहिए।
6. घरेलू अपशिष्ट, जैविक अपशिष्ट तथा रासायनिक अपशिष्टों को अधिकांशतः जला देना चाहिए। भस्मीकरण के फलस्वरूप अपशिष्ट पदार्थों का आयतन घट जाता है।
प्रश्न 14.
पीड़कनाशी तथा शाकनाशी से आप क्या समझते हैं? उदाहरण सहित समझाइए।
उत्तर:
पीड़कनाशी:
मुख्य रूप से संश्लेषित विषैले रसायन हैं जो पारिस्थितिकी प्रतिघाती हैं। पहले DDT का उपयोग फसलों में लगने वाले कीड़े-मकोड़ों की रोकथाम के लिए किया जाता था बाद में कीड़ों को DDT में प्रति, प्रतिरोधकता बढ़ गई । अतः दूसरे विषैले रसायन ऐल्ड्रीन और डाइएल्ड्रीन का उपयोग पीड़नाशी के रूप में होने लगा। लगभग सभी पीड़कनाशी जल में अघुलनशील तथा जैव अनिम्नीकरण होते हैं। आजकल जैव निम्नीकरण पदार्थ जो कार्बो-फॉस्फेट और कार्बामेट के नाम से उपयोग में लाए जाते हैं। परन्तु इनका उपयोग भी विषैला है तथा मानव जाति के लिए हानिकारक है। कृषि क्षेत्र में काम करने वाले लोग इसके शिकार हो चुके हैं।
प्रश्न 15.
हरित रसायन से आप क्या समझते हैं ? यह वातावरणीय प्रदूषण को रोकने में किस प्रकार सहायक है?
उत्तर:
हरित रसायन, रसायन विज्ञान तथा अन्य विज्ञानों के ज्ञात ज्ञान तथा सिद्धांतों के उपयोग की विधि है। जिससे पर्यावरण के दुष्प्रभावों को कम किया जा सके। हरित रसायन उत्पादन का प्रक्रम है जो पर्यावरण में न्यूनतम प्रदूषण या खराबी लाए। एक प्रक्रम में उत्पन्न होने वाले सह-उत्पादों को यदि लाभदायक तरीके से उपयोग नहीं किया जाए तो पर्यावरण प्रदूषण बढ़ाते हैं। ऐसे प्रक्रम न पर्यावरणीय दृष्टि से हानिकारक हैं, बल्कि महँगें भी हैं। उत्पादों के अपव्यय तथा इनका विसर्जन दोनों ही वित्तीय रूप से हानिकारक होते हैं। विकास कार्यों के साथ-साथ वर्तमान ज्ञान का रासायनिक हानि को कम करने के लिए उपयोग में लाना ही हरित रसायन का आधार है।”
प्रश्न 16.
क्या होता है, जब भू-वायुमण्डल में ग्रीन हाऊस गैसें नहीं होती ? विवेचना कीजिए।
उत्तर:
पृथ्वी की सतह से वापस विकरित सौर ऊर्जा को विभिन्न हरित गृह गैसों द्वारा अवशोषित कर लिया जाता है। इसके परिणामस्वरूप पृथ्वी का वायुमण्डल गर्म हो जाता है। यह वनस्पति की वृद्धि में सहायक होता है और जीवन का भी समर्थन करता है। इस प्रभाव की अनुपस्थिति में पृथ्वी की सतह पर पादप और जन्तु, दोनों का ही कोई जीवन नहीं होगा।
प्रश्न 17.
एक झील में अचानक असंख्य मृत मछलियाँ तैरती हुई मिली। इसमें कोई विषाक्त पदार्थ नहीं था, परन्तु बहुतायत में पादपप्लवक पाए गए। मछलियों के मरने का कारण बताइए।
उत्तर:
फाइटोप्लैंकटन में वृद्धि का कारण जल में पत्तियों, घास आदि जैसे कार्बनिक पदार्थ की उपस्थिति होती है। यह जल में घुलित बहुत-सी ऑक्सीजन का उपयोग कर लेते हैं, जो वास्तव में समुद्री जन्तुओं विशेष रूप से मछली के लिए बहुत आवश्यक है। यदि जल में घुलित ऑक्सीजन का स्तर 6 ppm से कम हो तो इसका तात्पर्य है कि यह ऑक्सीजन जल में रहने वाली विविध मछलियों के लिए पर्याप्त नहीं है। वे अन्ततः मर जाएँगी। हो सकता है कि इस स्थिति में भी ऐसा ही हुआ हो।
![]()
प्रश्न 18.
घरेलू अपशिष्ट किस प्रकार खाद के रूप में काम आ सकते हैं?
उत्तर:
घरेलू अपशिष्ट में जैव निम्नीय और जैव अनिम्नीय दोनों घटक होते हैं। जैव अनिम्नीय घटक में प्लास्टिक, काँच, धातु की छीलन आदि होता है तथा इसे पृथक् किया जा सकता है। जैव निम्नीय अंश, जो कि कार्बनिक पदार्थ से बना होता है, को उचित विधियों द्वारा खादों में रूपान्तरित किया जा सकता है।
प्रश्न 19.
आपने अपने कृषि-क्षेत्र अथवा उद्यान में कम्पोस्ट खाद के लिए गड्ढे बना रखे हैं। उत्तम कम्पोस्ट बनाने के लिए इस प्रक्रिया की व्याख्या दुर्गन्ध, मक्खियों तथा अपशिष्टों के चक्रीकरण के सन्दर्भ में कीजिए।
उत्तर:
उद्यानों में पौधों एवं घासों के स्वस्थ विकास के लिए समय-समय पर कम्पोस्ट खाद की आवश्यकता पड़ती है। स्थान उपलब्ध होने पर कम्पोस्ट खाद के गड्ढों को नजदीक में ही बनाते हैं। स्थान की कमी के कारण शहरी क्षेत्रों में इस सन्दर्भ में समस्या आ सकती है। इन गड्ढों से सामान्यतः दुर्गन्ध निकलती रहती है एवं मक्खियाँ भिनभिनाती रहती है। यह स्वास्थ्य के लिए बहुत बुरा होता है। इससे बचने के लिए गड्ढे को अच्छी तरह से ढंक देते हैं। काँच के सामान, प्लास्टिक के थैले, पुराने समाचार-पत्र आदि नियमित रूप से कबाड़ी को देना चाहिए। अन्ततः ये पुनर्चक्रीकरण संयंत्रों में भेज दिए जाते हैं, जिससे प्रदूषण की समस्या उत्पन्न नहीं होती है।
पर्यावरणीय रसायन अन्य महत्वपूर्ण प्रश्न अति
पर्यावरणीय रसायन वस्तुनिष्ठ प्रश्न
प्रश्न 1.
सही विकल्प चुनकर लिखिए –
प्रश्न 1.
प्रदूषण रहित ऊर्जा का स्रोत है –
(a) जीवाश्म ईंधन
(b) सूर्य
(c) गैसोलीन
(d) नाभिकीय ऊर्जा
उत्तर:
(b) सूर्य
![]()
प्रश्न 2.
कौन-से विकिरण O3, का निर्माण करते हैं –
(a) पराबैंगनी
(b) दृश्य प्रकाश
(c) अवरक्त
(d) रेडियो तरंगें
उत्तर:
(a) पराबैंगनी
प्रश्न 3.
कौन-से विकिरण हरित गृह प्रभाव देते हैं –
(a) अवरक्त
(b) दृश्य प्रकाश
(c) पराबैंगनी
(d) x-किरण।
उत्तर:
(a) अवरक्त
प्रश्न 4.
PAN उत्तरदायी है –
(a) ओजोन अपक्षय के लिये
(b) स्मॉग के लिये
(c) अम्ल वर्षा के लिये
(d) विषाक्त भोजन के लिये
उत्तर:
(b) स्मॉग के लिये
प्रश्न 5.
कौन-सा वायु प्रदूषक नहीं है –
(a) H2
(b) H2S
(c) NOx
(d) O3
उत्तर:
(a) H2
प्रश्न 6.
जेट विमानों से फ्लोरोकार्बन के रूप में निकलने वाला वायु प्रदूषक है –
(a) प्रकाश रासायनिक ऑक्सीकारक
(b) प्रकाश रासायनिक अपचायक
(c) ऐरोसॉल
(d) भौतिक प्रदूषक
उत्तर:
(c) ऐरोसॉल
प्रश्न 7.
अम्ल वर्षा में नहीं होता –
(a) H2SO4
(b) HNO3
(c) H2SO3
(d) CH3COOH
उत्तर:
(d) CH3COOH
![]()
प्रश्न 8.
फेफड़ों की बीमारी के लिये उत्तरदायी है –
(a) O2
(b) N2
(c) CO2
(d) SO2
उत्तर:
(d) SO2
प्रश्न 9.
O3 का निर्माण किसमें होता है –
(a) ट्रोपोस्फीयर
(b) स्ट्रेटोस्फीयर
(c) मेसोस्फीयर
(d) थर्मोस्फीयर
उत्तर:
(b) स्ट्रेटोस्फीयर
प्रश्न 10.
अम्ल वर्षा के लिये ‘सिंक’ है –
(a) पत्तियाँ
(b) जलाशय
(c) चूने का पत्थर
(d) CO2
उत्तर:
(c) चूने का पत्थर
प्रश्न 11.
प्राथमिक प्रदूषक है –
(a) SO3
(b) NO2
(c) N2O
(d) NO
उत्तर:
(d) NO
प्रश्न 12.
सबसे खतरनाक है –
(a) धुआँ
(b) धूल
(c) स्मॉग
(d) NO
उत्तर:
(c) स्मॉग
प्रश्न 13.
निम्न में कौन-सा हीमोग्लोबिन से तीव्रता से संयोग करता है –
(a) CO
(b) NO
(c) O2
(d) CO2
उत्तर:
(b) NO
प्रश्न 14.
ऐरोमैटिक यौगिक जो कणिका पदार्थ के रूप में पाया जाता है –
(a) बेंजीन
(b) टॉलूईन
(c) नाइट्रोबेंजीन
(d) बहुवलय हाइड्रोकार्बन
उत्तर:
(d) बहुवलय हाइड्रोकार्बन
प्रश्न 15.
ओजोन के क्षय से निम्न रोग होता है –
(a) रक्त कैंसर
(b) फेफड़े का कैंसर
(c) त्वचा कैंसर
(d) छाती कैंसर
उत्तर:
(c) त्वचा कैंसर
![]()
प्रश्न 2.
रिक्त स्थानों की पूर्ति कीजिए –
- जेट विमानों से ……………… फ्लोरोकार्बन के रूप में निकलने वाला प्रदूषक है।
- B.H.C. की तुलना में D.D.T. ………. खतरनाक प्रदूषक है।
- वायुमंडल के …………… स्तर में सर्वाधिक वायु प्रदूषक होते हैं।
- ओजोन परत ………….. किरणों से हमारा बचाव करती है।
- ……………… तथा …………. के ऑक्साइड अम्ल वर्षा कराते हैं।
- ……………. ओजोन परत क्षय का प्रमुख कारण है।
- वे घटक, जो प्रदूषण उत्पन्न करते हैं …………… कहलाते हैं।
- ……………. के कारण फेफड़ों पर हानिकारक प्रभाव पड़ता है।
- सल्फर डाइऑक्साइड प्रदूषक …………… की बीमारी के लिये उत्तरदायी हैं।
- पराबैंगनी प्रकाश …………… के लिये उत्तरदायी है।
उत्तर:
- ऐरोसॉल
- अधिक
- ट्रोपोस्फीयर
- पराबैंगनी
- नाइट्रोजन, सल्फर
- C.F.C.
- प्रदूषक
- प्रकाश रासायनिक स्मॉग
- फेफड़ों
- त्वचा कैंसर।
प्रश्न 3.
उचित संबंध जोडिए –

उत्तर:
- (b)
- (a)
- (d)
- (c)
- (g)
- (h)
- (e)
- (f).
प्रश्न 4.
एक शब्द/वाक्य में उत्तर दीजिए –
- दो वायु प्रदूषक के नाम लिखिए।
- दो प्रदूषकों के नाम बताइए जो ओजोन परत का क्षय करते हैं।
- ओजोन किस क्षेत्र में पायी जाती है?
- कोई दो ग्रीन हाऊस गैसों के नाम बताइए।
- P.A.N. क्या है?
- C.E.C. क्या है?
- दूषक क्या हैं?
- स्पेसियेशन क्या है?
- सिंक क्या है?
- पृथ्वी पर सबसे बड़े सिंक का नाम लिखिए।
- ग्रीन हाऊस प्रभाव क्या है?
- अम्ल वर्षा की क्रिया-विधि समझाइये।
- सन् 1984 में भोपाल त्रासदी में रिसने वाली गैस थी।
- ओजोन परत के क्षरण के लिये प्रमुख उत्तरदायी घटक का नाम क्या है?
- जल प्रदूषण के कारण होने वाली बीमारी का नाम लिखिए।
उत्तर:
- SO2, SO3
- (i) नाइट्रिक ऑक्साइड का चक्र (NO) तथा (ii) C.E.C. (क्लोरो- फ्लुओरोकार्बन) जिनमें फ्रीऑन प्रमुख है।
- समताप मण्डल में
- C.F.C. और CO2
- परॉक्सी-एसिल नाइट्रेट (PAN) है जो एक प्रकाश रासायनिक स्मॉग है।
- यह क्लोरो-फ्लुओरो कार्बन (C.F.C.) है, जो ओजोन परत के क्षय का मुख्य कारण है।
- ऐसे पदार्थ जो बाहर से अतिरिक्त रूप से पर्यावरण को दूषित करते हैं दूषक कहलाते हैं।
- एक ही तत्व से अनेक प्रदूषक निर्मित हो सकते हैं उनमें यह निर्धारण करना कि कौन-सा पदार्थ अधिक खतरनाक है, स्पेसियेशन कहलाता है।
- सिंक वह है जिसमें कोई वस्तु पूरी तरह से समा जाती है फिर भी सिंक पर कोई असर नहीं होता।
- पृथ्वी पर समुद्र से बड़ा कोई सिंक नहीं है।
- ऊष्मा विकिरणों (IR किरणें) का CO2 जैसी गैसों के कारण पृथ्वी की सतह पर ही बार-बार परावर्तित होकर तापमान में वृद्धि करना ग्रीन हाऊस प्रभाव कहलाता है।
- सभी प्रदूषक गैसों का धुएँ के रूप में विभिन्न प्रदूषण कण जीवाश्म तथा ईंधनों के जलने के कारण ही मुख्य रूप से फैलते हैं। कारखाने तथा अन्य इंजनों के अत्यन्त उच्च ताप के कारण N,तथा 0)के संयोग से नाइट्रोजन के ऑक्साइड भी वायुमंडल में फैलते हैं। ये विभिन्न गैसें वर्षा की बूंदों के रूप में पृथ्वी पर गिरती हैं, इसे अम्ल वर्षा कहते हैं।
- CH3-N = C = 0 (मेथिल आइसोसाइनेट)
- क्लोरो फ्लुओरो कार्बन
- कॉलरा, पेचिश, पीलिया इत्यादि।
पर्यावरणीय रसायन अति लघु उत्तरीय प्रश्न
![]()
प्रश्न 1.
पर्यावरणीय रसायन शास्त्र को परिभाषित कीजिए।
उत्तर:
पर्यावरणीय रसायन शास्त्र, विज्ञान की वह शाखा है, जिसमें हमारे वातावरण पर रसायनों के परिणाम (जैसे-उत्पत्ति, परिवहन, अभिक्रियाएँ, प्रभाव तथा तथ्यों आदि) का अध्ययन किया जाता है।
प्रश्न 2.
ग्रीन हाऊस प्रभाव के कारण भूमण्डलीय ताप में वृद्धि हो रही है। ग्रीन हाऊस प्रभाव के लिए कौन-से पदार्थ उत्तरदायी हैं?
उत्तर:
ग्रीन हाऊस गैसें तथा कार्बनमोनोऑक्साइड, मेथेन, नाइट्रस ऑक्साइड, ओजोन तथा क्लोरोफ्लु ओरोकार्बन (C.E.C.S.) ग्रीन हाऊस प्रभाव के लिए उत्तरदायी हैं। ये गैसें पृथ्वी की सतह से जाने वाली विकिरणों को अवशोषित करके पृथ्वी का तापमान नियंत्रित करती हैं।।
प्रश्न 3.
ओजोन एक विषैली गैस तथा प्रबल ऑक्सीकारक अभिकर्मक है फिर भी ऊपरी समतापमण्डल में इसकी उपस्थिति अति महत्वपूर्ण है। यदि इस क्षेत्र से ओजोन को पूर्णतया हटा लिया जाए तो क्या होगा?
उत्तर:
ओजोन सूर्य की हानिकारक पराबैंगनी किरणों को पृथ्वी की सतह तक पहुँचने से रोकती है जिसके कारण यह पराबैंगनी किरणों के दुष्प्रभाव से जन-जीवन की रक्षा करती है। यदि ऊपरी समतापमण्डल से ओजोन को पूर्णतया हटा लिया जाए तो पराबैंगनी किरणें पृथ्वी पर पहुँच जाएगी तथा अनेकों बीमारियाँ जैसेत्वचा झुलसना, त्वचा कैंसर आदि बीमारियाँ उत्पन्न हो जाएंगी।
प्रश्न 4.
जल में विलेय ऑक्सीजन के स्रोत क्या हैं?
उत्तर:
जल में विलेय ऑक्सीजन के स्रोत निम्न हैं –
- प्रकाश-संश्लेषण
- प्राकृतिक ऐरेशन
- कृत्रिम ऐरेशन।
प्रश्न 5.
जल में घुली हुई ऑक्सीजन जलीय जीवन के लिए अत्यन्त महत्वपूर्ण है। जल में घुली ऑक्सीजन की कमी के लिए कौन-से कारक उत्तरदायी है?
उत्तर:
जल में घुलित ऑक्सीजन की कमी के लिए, फॉस्फेटी तथा नाइट्रेट उर्वरकों, अपमार्जक, मानवजन्तु मल का बहाव तथा भोजन, कागज, लुग्दी उद्योगों द्वारा कार्बनिक अपशिष्टों आदि का जल में अत्यधिक उत्सर्जन है। सूक्ष्मजीव जो कार्बनिक पदार्थों का ऑक्सीकरण करते हैं, वे भी जल में विलेय ऑक्सीजन का प्रयोग करते हैं। इसके साथ-साथ रात में प्रकाश-संश्लेषण तो रुक जाता है परन्तु जलीय जीव तथा पादप निरंतर श्वसन करते हैं, जिसके कारण जल में घुलित ऑक्सीजन की मात्रा में कमी आती है।
प्रश्न 6.
जैवनिम्नीकृत तथा अजैवनिम्नीकृत प्रदूषक कौन-कौन से हैं?
उत्तर:
जैवनिम्नीकृत प्रदूषक-जीवाणुओं द्वारा अपघटित हो सकने वाले पदार्थ हैं। उदाहरण-मल, पशुओं का गोबर, फल-सब्जियों के छिलके आदि। अजैवनिम्नीकृत प्रदूषक-जीवाणुओं द्वारा अपघटित न हो सकने वाले पदार्थ हैं। उदाहरण-मर्करी, लेड, डी.डी.टी. काँच एवं प्लास्टिक आदि।
![]()
प्रश्न 7.
प्रदूषण किसे कहते हैं?
उत्तर:
संतुलित वातावरण में प्रत्येक घटक लगभग एक निश्चित मात्रा एवं अनुपात में रहता है। कभीकभी वातावरण में एक अथवा अनेक घटकों की मात्रा या तो आवश्यकता से अधिक बढ़ जाती है अथवा वातावरण में हानिकारक घटकों का प्रवेश हो जाता है जो जीवधारियों के लिये किसी-न-किसी रूप में हानिकारक सिद्ध होता है, प्रदूषण कहलाता है।
प्रश्न 8.
प्रदूषक किसे कहते हैं?
उत्तर:
ऐसे पदार्थ जिनकी मात्रा पर्यावरण में आवश्यकता से अधिक होने लगे तथा एक सीमा से अधिक बढ़ जाने के कारण मानव को, प्राणी जगत को या वनस्पति जगत को नुकसान पहुँचाने लगे, वे प्रदूषक कहलाते हैं। ये पदार्थ मुख्य रूप से निम्नलिखित हो सकते हैं – CO, CO2, NO, NO2, SO, इत्यादि।
प्रश्न 9.
दूषक किसे कहते हैं?
उत्तर:
कुछ पदार्थ ऐसे होते हैं, जो प्रकृति में तो नहीं पाये जाते किन्तु मानव द्वारा कृत्रिम रूप से निर्मित किये जाते हैं। ये पदार्थ नष्ट हुये बगैर पर्यावरण को दूषित करते हैं।
प्रश्न 10.
ओजोन पर्त क्षरण के लिये उत्तरदायी गैसों या रसायन के नाम लिखिए।
उत्तर:
ओजोन पर्त के क्षरण के लिये वायुमण्डल में उपस्थित नाइट्रिक ऑक्साइड, परमाणवीय ऑक्सीजन तथा क्लोरोफ्लुओरो कार्बन उत्तरदायी हैं।
प्रश्न 11.
ग्रीन हाऊस गैसें क्या हैं?
उत्तर:
CO2 ओजोन व जलवाष्प को ग्रीन हाऊस गैसें कहा जाता है। इनमें अवरक्त विकिरणों को अवशोषित करने का गुण होता है। अतः इन्हें ग्रीन हाऊस गैसें कहा जाता है।
![]()
प्रश्न 12.
प्रदूषित वायु किसे कहते हैं?
उत्तर:
यदि वायु में ऐसे पदार्थ उपस्थित हो जो जीवों के स्वास्थ्य पर बुरा प्रभाव डालें तो उस वायु को प्रदूषित वायु कहते हैं। प्रदूषित वायु में मुख्यत: CO2, SO2, SO3, CO निलम्बित अणु रहते हैं।
प्रश्न 13.
अंटार्कटिका पर ही ओजोन का क्षय होता है, क्यों?
उत्तर:
क्योंकि समतापमण्डल के दूसरे भागों में मुक्त क्लोरीन अणुओं में बदल जाते हैं, जबकि अंटार्कटिका पर उत्पन्न यौगिक क्लोरीन मुक्त मूलकों से घिर जाते हैं जिससे ओजोन का क्षय होता है।
प्रश्न 14.
जल के किसी नमूने के BOD मापन की क्या उपयोगिता है?
उत्तर:
BOD जल के किसी दिए गए नमूने में उपस्थित कार्बनिक जैवनिम्नीकृत पदार्थ द्वारा जनित प्रदूषण का मापन है। BOD का कम मान संकेत करता है कि जल में कार्बनिक अपशिष्ट कम मात्रा में उपस्थित है।
प्रश्न 15.
सल्फर डाइऑक्साइड का उत्प्रेरक की अनुपस्थिति में सल्फर ट्राइऑक्साइड में ऑक्सीकरण एक मंद परिवर्तन है परन्तु इसका ऑक्सीकरण वायुमण्डल में शीघ्रता से होता है बताइये कि ऐसा क्यों होता है? SO2 में SO3, के परिवर्तन की रासायनिक समीकरण भी दीजिए।
उत्तर:
SO2 से SO3 के ऑक्सीकरण में, प्रदूषित वायु में उपस्थित कणिकीय द्रव्य उत्प्रेरक की भाँति कार्य करते हैं।
![]()
प्रश्न 16.
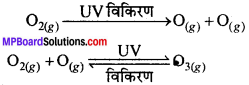
समतापमण्डल में ओजोन किस प्रकार निर्मित होती है?
उत्तर:
समतापमण्डल में ओजोन पराबैंगनी विकिरणों की डाइऑक्सीजन (O2) अणु पर क्रिया से उत्पन्न होती है। पराबैंगनी किरणें ऑक्सीजन अणु को ऑक्सीजन परमाणु में अपघटित कर देती है। ये ऑक्सीजन परमाणु, ऑक्सीजन अणु के साथ संयुग्मित होकर ओजोन अणु बनाते हैं।
प्रश्न 17.
क्लोरोसीन क्या है?
उत्तर:
पौधों में क्लोरोफिल पदार्थ धीमी गति से बनता है। यह SO की उपस्थिति के कारण होता है। यह प्रदूषण क्लोरोसीन कहलाता है।
प्रश्न 18.
मैटाथेसीज क्या है?
उत्तर:
मैटाथेसीज उस विज्ञान का नाम है, जिसमें वैज्ञानिक विधियों का सामान्य आदमी के लिए क्या उपयोग है, का अध्ययन किया जाता है।
![]()
प्रश्न 19.
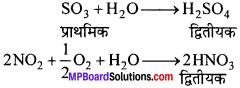
प्राथमिक एवं द्वितीयक वायु प्रदूषक किसे कहते हैं?
उत्तर:
प्राथमिक वायु प्रदूषक-जो वायु प्रदूषक प्राकृतिक या मानवीय कारणों से सीधे वायु को प्रदूषित करते हैं, प्राथमिक वायु प्रदूषक कहलाते हैं। जैसे – CO, NO, CO2, SO2.
द्वितीयक वायु प्रदूषक-ऐसे वायु प्रदूषक जो वायुमण्डल में ही प्रदूषकों के बीच रासायनिक क्रियाओं से या गैसों तथा प्रदूषकों के बीच क्रियाओं से बनते हैं, उन्हें द्वितीयक प्रदूषक कहते हैं। जैसे –
प्रश्न 20.
प्रकाश रासायनिक स्मॉग किसे कहते हैं?
उत्तर:
स्मॉग का अर्थ धुएँ और कोहरे का सम्मिश्रण है जब इस स्मॉग में ऑक्सीकारकों की उच्च सान्द्रता उपस्थित हो तो इसे प्रकाश रासायनिक स्मॉग कहते हैं। धुआँ एक वायु प्रदूषक है जिसमें ईंधन पदार्थों के अपूर्ण दहन के कारण अनेक ठोस कण वायुमण्डल में निलम्बित रहते हैं। गैसीय माध्यम में अति सूक्ष्म आकार के ठोस कणों के विक्षेपण से उत्पन्न बादल को वायु विलय कहते हैं और धुएँ व कोहरे के संयोग से धुन्ध बनती है।
प्रश्न 21.
अम्लीय वर्षा किसे कहते हैं?
उत्तर:
वायु प्रदूषण के कारण वायुमण्डल में विभिन्न गैसों जैसे – CO2, SO2, SO3, NO2, इत्यादि की सान्द्रता में वृद्धि होती है। ये विभिन्न गैसें वर्षा की बूंदों में अपनी-अपनी विलेयता के अनुसार विलेय होकर अम्ल बनाती हैं। वर्षा के रूप में गिरने वाली ये बूंदें ही अम्लीय वर्षा कहलाती हैं।
CO2 + H2O → H2CO3
SO3 + H2O → H2SO4
प्रश्न 22.
CO2 कब प्रदूषक कहलाती है?
उत्तर:
वायुमण्डल में CO2 की सामान्य मात्रा किसी प्रकार भी हानिकारक नहीं है। बल्कि जीव एवं पेड़-पौधे इससे अपने भोजन का निर्माण करते हैं। लेकिन जब विभिन्न प्रक्रियाओं के कारण जब वायुमण्डल में इसकी सान्द्रता आवश्यकता से अधिक हो जाती है तो प्राकृतिक संतुलन को बिगाड़ कर नुकसान पहुँचाती है।
प्रश्न 23.
ग्रीन हाऊस प्रभाव के लिये कार्बन डाइऑक्साइड को अधिक उत्तरदायी समझा जाता है, क्यों?
उत्तर:
जलवाष्प केवल पृथ्वी के अत्यधिक निकट पायी जाती है तथा ओजोन वायुमण्डल के ऊपरी भाग में पायी जाती है। इनकी तुलना में CO2 वायुमण्डल में समान रूप से सभी स्थान में पायी जाती है। इसलिये ग्रीन हाऊस प्रभाव के लिये CO2 अधिक उत्तरदायी है क्योंकि CO2 में सूर्य के अवरक्त प्रकाश की किरणों को अवशोषित करने की क्षमता होती है। इसके फलस्वरूप वातावरण में ग्रीन हाऊस प्रभाव उत्पन्न होता है।
प्रश्न 24.
अम्ल वर्षा को ताजमहल के लिए हानिकारक माना जाता है, क्यों?
उत्तर:
ताजमहल संगमरमर (CaCO3) से बना है। अम्लीय वर्षा में H2SO4 होता है, जो बहुत ही तनु अवस्था में होता है, जो संगमरमर से क्रिया करता है। अतः संगमरमर धीरे-धीरे क्षय हो जाता है।
CaCO3(aq) + H2SO4(aq) → CaSO4(aq) + CO2(g) + H2O(l)
![]()
प्रश्न 25.
एक व्यक्ति नगर निगम द्वारा सप्लाई किए जल का उपयोग करता है। जल की कमी के कारण भू-जल का प्रयोग प्रारंभ कर देता है। उसे विरेचकता का अनुभव होता है। इसके क्या कारण हो सकते हैं?
उत्तर:
विरेचकता का अनुभव केवल तभी हो सकता है जब जल में उपस्थित सल्फेट की सान्द्रता 500 ppm से अधिक हो। अन्यथा माध्यमिक स्तर पर जल हानिरहित होता है।
प्रश्न 26.
फ्लाई ऐश प्रदूषण किसे कहते हैं?
उत्तर:
कोल में उपस्थित लाइम स्टोन राख में CaO के रूप में रहता है तथा राख के साथ धूल में उड़ सकता है। वातावरण में स्थित H2SO4 की सूक्ष्म बूंदें CaO से क्रिया कर CaSO4 के कण बना देती है। हवा में उपस्थित अमोनिया भी H2SO4 के साथ क्रिया कर (NH4)2SO4 के कणों में बदल जाता है। राख का हवा के साथ उड़कर प्रदूषण उत्पन्न करना फ्लाई ऐश प्रदूषण कहलाता है।
प्रश्न 27.
संक्रमित जल किसे कहते हैं? संक्रमित जल से होने वाली बीमारियों के नाम लिखिए।
उत्तर:
वह जल जिसमें प्रदूषण के कारण जीवों के विविध हानिकारक पदार्थ तथा सूक्ष्म जीव उपस्थित हैं, संक्रमित जल कहलाता है। संक्रमित जल से होने वाली बीमारियाँ निम्नलिखित हैं –
- हैजा
- पीलिया
- पेचिश
- चर्म रोग।
प्रश्न 28.
ग्लोबल वॉर्मिंग से क्या समझते हो?
उत्तर:
CO2 मेथेन, क्लोरो फ्लुओरो कार्बन आदि गैसों की वायुमण्डल में अधिक मात्रा में उपस्थिति के कारण सूर्य की गर्मी रुक जाने से पृथ्वी के तापमान में वृद्धि ग्लोबल वॉर्मिंग कहलाती है। ग्लोबल वॉर्मिंग ग्रीन हाऊस का दुष्परिणाम है।
प्रश्न 29.
आयनोस्फीयर किसे कहते हैं?
उत्तर:
पृथ्वी की सतह से 50 से 90 कि.मी. की ऊँचाई तक लगभग 40 कि.मी. की मोटाई वाली पर्त मेसोस्फीयर कहलाती है। इस क्षेत्र में सभी गैसें आयनित अवस्था में रहती हैं इसलिये इसे आयनोस्फीयर भी कहते हैं।
O2 + O+ → O2+ + O
O+ + N2 → NO+ + N
N2+ + O2 → N2 + O2+
पर्यावरणीय रसायन लघु उत्तरीय प्रश्न
प्रश्न 1.
कार्बन मोनोऑक्साइड हमारे शरीर पर क्या बुरा प्रभाव डालती है?
उत्तर:
कार्बन मोनोऑक्साइड का हीमोग्लोबिन पर अवशोषण ऑक्सीजन की तुलना में अधिक होता है। यह कार्बन मोनोऑक्साइड लाल वर्णक हीमोग्लोबिन से संयोग कर ज्यादा स्थायी यौगिक कार्बोक्सी हीमोग्लोबिन बनाता है जो स्थायी होने के कारण कोशिकाओं में पहुँचकर हीमोग्लोबिन को मुक्त नहीं करता है, अतः रक्त की ऑक्सीजन संवहन क्षमता कम हो जाती है। इसके कारण मामूली सिर दर्द, सुस्ती से लेकर अंत में मृत्यु तक हो सकती है।
प्रश्न 2.
सल्फर डाइऑक्साइड के दुष्प्रभाव लिखिए।
उत्तर:
SO2 के दुष्प्रभाव:
- SO2 श्वसन नली एवं फेफड़ों को नुकसान पहुँचाती है। इस कारण फेफड़ों की कई बीमारी, कैंसर आदि हो सकता है।
- यह पेड़-पौधों की वृद्धि को भी रोकती है। इसके कारण हरी पत्तियों का पीला पड़ना क्लोरोसिस कहलाता है।
- SO2 के कारण होने वाली अम्ल वर्षा कीमती इमारती पत्थरों को नुकसान पहुँचाती है।
- SO2 गैस कोरोसन (Corrosion) को बढ़ावा देती है।
![]()
प्रश्न 3.
नाइट्रोजन ऑक्साइड के दुष्प्रभाव लिखिए।
उत्तर:
नाइट्रोजन ऑक्साइड के दुष्प्रभाव:
- वातावरण में NO, NO2 आदि गैसों की आवश्यकता से अधिक उपस्थिति आँखों में जलन उत्पन्न करती है।
- इसकी अधिकता से फेफड़ों एवं हृदय संबंधी रोग हो जाते हैं।
- नाइट्रोजन के ऑक्साइड धातुओं में संक्षारण को बढ़ा देते हैं।
- प्रकाश संश्लेषण की दर को कम करता है।
- रंजकों के चमक को कम कर देता है।
प्रश्न 4.
ग्रीन हाऊस प्रभाव के कुप्रभाव क्या हैं?
उत्तर:
ग्रीन हाऊस प्रभाव का मुख्य कारण वायुमण्डल में CO2 की अधिक मात्रा में उपस्थिति है। जो अवरक्त प्रकाश को अवशोषित कर वातावरण का ताप बढ़ाने में सहायक होती है। इस प्रकार ताप में वृद्धि के कारण मौसम में बदलाव आयेगा। सूर्य की किरणें कैंसर जैसे भयानक रोग को जन्म देती हैं। कहीं सूखा पड़ेगा, कहीं गर्म हवायें चलेंगी, कहीं भीषण तूफान एवं कहीं बाढ़ आयेगी। आर्कटिका एवं अंटार्कटिका के विशाल हिमखण्ड पिघल जायेंगे जिससे समुद्र के जल स्तर में वृद्धि हो जायेगी जिसके कारण समुद्र तटीय नगर समुद्र में डूब जायेंगे।
प्रश्न 5.
स्मॉग क्या है? इसके प्रकार लिखते हुये इसकी क्रियाविधि को समझाइये।
उत्तर:
धुएँ तथा कोहरे का सम्मिश्रण स्मॉग कहलाता है।
स्मॉग के प्रकार:
- रासायनिक स्मॉग – कोहरे के साथ मिली SO2 गैस का धुएँ से संयोग रासायनिक स्मॉग को जन्म देती है।
- प्रकाश रासायनिक स्मॉग – जहाँ तेज धूप पड़ती है वहाँ प्राथमिक वायु प्रदूषक प्रकाश रासायनिक क्रिया द्वारा द्वितीय प्रदूषक निर्मित करते हैं। इनकी प्रकृति ऑक्सीकारक होती है। जहाँ प्रदूषक गैस अधिक हो हवा रुकी हुई है, वहाँ धुएँ के साथ स्मॉग निर्मित होता है।
क्रियाविधि:
- सूर्योदय से पहले ऑटोमोबाइल इंजन का प्रयोग किया जा रहा हो तो मुख्य रूप से CO तथा NO गैस निकलती है। यह वायु के संपर्क में भूरे रंग की NO2 गैस बनाती है।
2NO + O2 → 2NO2 - पराबैंगनी किरणों के प्रभाव से NO2 द्वारा क्रियाशील ऑक्सीजन मुक्त होती है।
2NO + O2 → 2NO2 - यह क्रियाशील ऑक्सीजन इंजन से निकलने वाले हाइड्रोकार्बन को मुक्त मूलक में बदल देती है।
- ये कार्बनिक मुक्त मूलक कई शृंखला अभिक्रियाओं द्वारा अवांछित यौगिकों का निर्माण करते हैं और ये प्रदूषक ही स्मॉग का निर्माण करते हैं।
![]()
प्रश्न 6.
जल प्रदूषण के कारणों का उल्लेख कीजिए।
उत्तर:
- बड़े-बड़े कारखानों के अपशिष्ट पदार्थों को समुद्रों, नदियों या झीलों में फेंका जाता है।
- खेतों में तरह-तरह के उर्वरक, खाद तथा कीटनाशकों का प्रयोग किया जाता है। ये सभी पदार्थ खेतों की मिट्टी में मिश्रित हो जाते हैं तथा वर्षा के जल में घुलकर नदियों तथा जलाशय में गिरते हैं।
- परमाणु विद्युत् केन्द्र जल को प्रदूषित करते हैं।
- जनसंख्या में वृद्धि के कारण नदियों के किनारे बस्तियाँ बन गई हैं इनके घर से निकलने वाले अपशिष्ट जल को प्रदूषित करते हैं।
- ग्रामीण क्षेत्रों में तालाब में लोग नहाते हैं, कपड़े धोते हैं जो जल को प्रदूषित करते हैं।
प्रश्न 7.
कृत्रिम ग्रीन हाऊस का निर्माण कैसे कर सकते हैं?
उत्तर:
काँच में यह गुण होता है कि वह दृश्य प्रकाश की किरणों को तो लगभग पूरी तरह अपवर्तित कर देता है परन्तु अधिक तरंग लंबाई के कारण IR किरणों को अपवर्तित कम तथा परावर्तित अधिक करता है। अतः काँच के अंदर उपस्थित IR विकिरणों का हिस्सा जो पृथ्वी से परावर्तित कर दिया जाता है। कुछ मात्रा में काँच द्वारा परावर्तित हो जाता है। इस प्रकार काँच द्वारा घिरा भाग गर्म होने लगता है।
प्रश्न 8.
जल प्रदूषण के कुप्रभाव लिखिए तथा इसका नियंत्रण कैसे करते हैं?
उत्तर:
जल प्रदूषण के कुप्रभाव:
- प्रदूषित जल के सेवन से हैजा, पीलिया, अतिसार, टायफाइड जैसे संक्रामक रोगों का प्रसार होता है।
- वाहितमल के कारण प्रदूषित जल में विलेय ऑक्सीजन की मात्रा घट जाती है, जिस कारण जलीय प्राणियों की मृत्यु हो जाती है।
- साबुन, डिटर्जेन्ट अपशिष्ट के कारण प्रदूषित जल मछलियों के लिये जानलेवा होता है।
नियंत्रण:
- वाहितमल को सीधे नदियों जलाशय में न फेंककर रासायनिक अभिक्रिया द्वारा खाद में परिवर्तित कर दिया जाये।
- समुद्र के नीचे परमाणु परीक्षण पर रोक लगाना चाहिये।
- पुनः चक्रण (Recycling) तरीकों द्वारा गोबर या मलमूत्र जैसे पदार्थों को उपयोगी पदार्थ में परिवर्तित किया जा सकता है।
प्रश्न 9.
मृदा प्रदूषण क्या है ? मृदा प्रदूषण पर किस प्रकार नियंत्रण किया जा सकता है?
उत्तर:
मृदा प्रदूषण:
मृदा के भौतिक, रासायनिक या जैविक गुणों में ऐसा कोई भी अवांछित परिवर्तन जिनका हानिकारक प्रभाव मनुष्य तथा अन्य जीवों पर पड़ता है या मृदा या भूमि की प्राकृतिक गुणवत्ता तथा उपयोगिता नष्ट हो जाये मृदा प्रदूषण कहलाता है।
नियंत्रण:
- औद्योगिक अपशिष्टों को कारखानों में उपचारित करके ही भूमि में डालना चाहिए।
- उर्वरक, कीटनाशी, कवकनाशी दवाओं का कम-से-कम प्रयोग करना चाहिए।
- मृदा अपरदन को रोकने के लिये वृक्षों का कटाव रोकना चाहिए जिससे भूमि की पर्त नष्ट न हो।
![]()
प्रश्न 10.
ओजोन पर्त के क्षय की क्रियाविधि को समझाइये।
उत्तर:
1. नाइट्रोजन ऑक्साइड के द्वारा क्षय-स्ट्रेटोस्फीयर में कुछ मात्रा में NO उपस्थित है। यह परमाणवीय ऑक्सीजन से संयोग कर NO बनाता है।
N2O + O → 2NO
NO2 गैस O3 से संयोग कर NO2 बनाती है।
NO + O3 → NO2 + O2
NO2 परमाणवीय ऑक्सीजन से टूटकर पुनः NO देता है।
NO2 + O → NO + O2
यह NO का चक्र O3 के क्षय का कारण बनता है।
2. C.F.C. के द्वारा क्षय-प्रकाशीय विघटन द्वारा यह मुक्त मूलक उत्पन्न करता है।

मुक्त मूलकों का निर्माण ओजोन पर्त का निरंतर क्षय कर रहा है।
पर्यावरणीय रसायन दीर्घ उत्तरीय प्रश्न
प्रश्न 1.
वायुमण्डल को कितने भागों में बाँटा गया है? प्रत्येक क्षेत्र में होने वाली रासायनिक क्रियाओं को समझाइये।
उत्तर:
वायुमण्डल को चार भागों में बाँटा गया है –
- ट्रोपोस्फीयर
- स्टेट्रोस्फीयर
- मेसोस्फीयर
- थर्मोस्फीयर।
1. ट्रोपोस्फीयर:
पृथ्वी की सतह से 8 से 12 कि.मी. की ऊँचाई तक का क्षेत्र ट्रोपोस्फीयर कहलाता है। इसके मुख्य घटक N2, O2, CO2, तथा H2O हैं। जीवाश्म ईंधन ऑक्सीजन में जलकर CO2 बनाता है।
CH4 + 2O2 → CO2 + 2H2O ट्रोपोस्फीयर में विभिन्न जीवाणुओं द्वारा यौगिक का विघटन होता है।
जीवाणु यौगिक + O2 → CO2 + H2O प्रकाश संश्लेषण भी इसी पर्त में होती है।
![]()
2. स्ट्रेटोस्फीयर:
10 से 50 किमी तक की लगभग 40 कि. मी. मोटाई की पर्त स्ट्रेटोस्फीयर कहलाती है।
(a) ऑक्सीजन का प्रकाश रासायनिक अपघटन
O2 \(\underrightarrow { hv } \) O + O
O + O + O \(\underrightarrow { hv } \) O2 + O*
उत्तेजित ऑक्सीजन अणु O2 दृश्य प्रकाश क्षेत्र में 636, 630, 558nm तरंग लंबाई वाले विकिरण उत्सर्जित करता है। इसे वायु दीप्त कहते हैं।
O* \(\underrightarrow { hv } \) O+ + e–
ऑक्सीजन परमाणु उत्तेजित होकर इलेक्ट्रॉन त्याग सकता है, जिससे धनावेशित ऑक्सीजन आयन बनता है।
O2 + O3 → O2 + O
पराबैंगनी किरणों से O3 का अणु पुनः विघटित हो जाता है।
O3 \(\underrightarrow { hv } \) O2 + O
O3 + O → O2 + O2
ओजोन को स्थायित्व प्रदान करने के लिये N2, O2 जैसा कोई दूसरा आवश्यक है, जो विकिरणों की अतिरिक्त ऊर्जा को अवशोषित कर O, के अपघटन को रोकता है।
O + O2, + M → O3 + M
इसके विपरीत स्ट्रेटोस्फीयर में NO या OH जैसे स्पीशीज हैं, तो वे Oz को विघटित कर देती हैं।
O3 + OH → O2 + HOO
O3 + NO → NO2 + O2
NO2 + O → NO + O2
N2 अणु स्वतः विकिरणों द्वारा अपघटित होता है तथा O2 से संयोग कर NO बनाता है।
N2 \(\underrightarrow { hv } \) N + N
O2 + N → NO + O
3. मेसोस्फीयर:
पृथ्वी की सतह से 50 से 90 कि.मी. की ऊँचाई तक लगभग 40 कि.मी. मोटाई वाली पर्त मेसोस्फीयर है।
O2 + O → O2 + O
O+ + N2 → NO+ + N
N2+ + O2 → N2 + O2+
4. थर्मोस्फीयर:
पृथ्वी की सतह से 90 कि.मी. की ऊँचाई से वायुमण्डल की यह पर्त प्रारम्भ होती है। 500 कि.मी. ऊँचाई तक का यह क्षेत्र थर्मोस्फीयर कहलाता है।
![]()
प्रश्न 2.
पार्टिकुलेट कितने प्रकार के होते हैं? वर्णन कीजिए।
उत्तर:
वातावरण में अत्यंत सूक्ष्म कण भी प्रदूषण के कारण हैं। विभिन्न प्रकार के कण तथा उनके स्रोत निम्नलिखित हैं –
1. कालिख:
कार्बन युक्त ईंधन के अपूर्ण ज्वलन से धुएँ के साथ कार्बन के सूक्ष्म कण वातावरण में फैल जाते हैं। इस प्रकार का दहन प्रायः सभी जगह होता रहता है।
2. धूल:
प्राकृतिक तथा मानवीय स्रोतों में ज्वालामुखी के फूटने के कारण, आँधी के कारण, खनन कार्य के कारण, वाहनों के चलने के कारण धूल वातावरण में फैलती है।
3. धातुओं के कण:
सीसा, पारा, Cr, As, Zn, Fe, Ni, Cd धातुओं के कण विभिन्न प्रकार से धातु उपचार के कारण वातावरण में फैलते हैं। इनकी कम मात्रा भी अत्यन्त हानिकारक है।
4.धातु ऑक्साइड के कण:
ईंधन में उपस्थित धातु दहन के पश्चात् ऑक्साइड बनाती है जिनके कण वातावरण में फैल जाते हैं। प्राकृतिक तेल में उपस्थित V2O5 के कण कोल के दहन Fe3O4 के कण मुख्य रूप से फैल जाते हैं।
5. फ्लाई ऐश प्रदूषण:
कोल में उपस्थित लाइम स्टोन राख में CaO के रूप में बच जाता है। राख के साथ धूल में उड़ सकता है। वातावरण में स्थित H2SO4 की सूक्ष्म बूंदें CaO से क्रिया कर CaSO4 के कण बना देती है। हवा में उपस्थित NH3 भी H2SO4 के साथ संयोग कर (NH4)2SO4 के कणों में बदल जाती है। राख हवा के साथ उड़कर सूक्ष्म कणों में बदलकर प्रदूषण उत्पन्न करती है।
6. एस्बेस्टस कण:
उद्योग तथा कारखानों से निकलने वाले धुएँ में उपस्थित एस्बेस्टस के कण प्रदूषण उत्पन्न करते हैं।
7. P.A.H. तथा T.E.L.:
PAH – पॉलीसाइक्लिक ऐरोमैटिक हाइड्रोकार्बन, TEL – टेट्राएथिल लेड है। जीवाश्म ईंधन तथा पेट्रोलियम के अपूर्ण दहन से P.A.H. सूक्ष्म कणों के रूप में प्रदूषण उत्पन्न करते हैं। नोदन को कम करने के लिए T.E.L. का उपयोग करते हैं। T.E.L. का दहन के पश्चात् PbO में ऑक्सीकरण होता है तथा इसमें C2H4Br2 तथा C2H4Cl मिलाया जाता है। यह PbO को PbBr2 व PbCl3 में परिवर्तित कर देता है। इस प्रकार T.E.L. का प्रदूषण लैड हैलाइड के रूप में होता है।
प्रश्न 3.
अम्ल वर्षा क्या होती है? पर्यावरण पर इससे होने वाले दुष्प्रभावों का विस्तार से वर्णन कीजिए।
उत्तर:
वायुमण्डल में अनेक प्रकार के गैसीय ऑक्साइड उपस्थित रहते हैं। वायु प्रदूषण के कारण वायु में इनकी % मात्रा में वृद्धि हो रही है। जब वर्षा का जल नीचे आता है तो ये ऑक्साइड जल में विलेय होकर अम्ल बनाते हैं तथा पृथ्वी पर वर्षा की बूंदों के रूप में गिरते हैं इसे अम्लीय वर्षा कहते हैं।
CO2 + H2O → H2CO3
SO3 + H2O → H2SO4
अम्ल वर्षा के प्रभाव:
- अम्ल वर्षा जमीन में उपस्थित क्षार का उदासीनीकरण कर देती है जिसके फलस्वरूप मिट्टी अम्लीय हो जाती है। इससे जीवाणुओं, पौधों और नाइट्रोजन स्थिरीकरण पर प्रभाव पड़ता है।
- अम्लीय वर्षा से नदियों, सरोवरों का जल अम्लीय हो जाता है जिससे इनमें रहने वाले जीव-जन्तुओं पर हानिकारक प्रभाव पड़ता है।
- अम्लीय वर्षा के कारण वन संपदा तथा वनस्पतियों का क्षय हो रहा है जिससे पतझड़ शीघ्र आ रहा है और पौधे सूख रहे हैं।
- अम्लीय वर्षा के कारण मूर्तियाँ तथा भवन अपनी चमक खो रहे हैं क्योंकि उसमें उपस्थित धातुओं का क्षरण होता है। कोटिंग भी नष्ट होती है। आगरा के ताजमहल पर अम्लीय वर्षा के कारण संगमरमर का क्षय हो रहा है।
- अम्ल वर्षा का जल पौधों की पत्तियों को हानि पहुँचाकर प्रकाश संश्लेषण की दर को मंद कर देता है।
- अम्ल वर्षा द्वारा त्वचा, फेफड़े तथा गले पर हानिकारक प्रभाव पड़ता है।
अम्ल वर्षा का नियंत्रण:
- नदियों तथा तालाबों को अम्लीय वर्षा से नहीं बचाया जा सकता है पर इसमें चूना मिलाकर इसके प्रभाव को कम किया जा सकता है।
- खेतों में भी चूना डालने से अम्लीय वर्षा के प्रभाव को कम किया जा सकता है।
- कम गंधक युक्त कोयले या लेड रहित पेट्रोल का उपयोग करने से तथा संयंत्रों में ईंधन के दहन की प्रक्रिया को नियंत्रित करके अम्लीय वर्षा के प्रभाव को नष्ट किया जा सकता है।
- अम्ल प्रदूषण करने वाली गैस तथा अधजले हाइड्रोकार्बन को प्रभावहीन यौगिक में परिवर्तित करके इसके प्रभाव को नियंत्रित करने का प्रयास किया जा रहा है।
प्रश्न 4.
सल्फर तथा नाइट्रोजन के ऑक्साइडों की प्रदूषण प्रक्रिया समझाइये। इन पर नियंत्रण किस प्रकार किया जा सकता है?
उत्तर:
नाइट्रोजन के ऑक्साइड:
NO प्राथमिक प्रदूषक है जबकि N2O तथा NO2 अन्य ऑक्साइड द्वितीयक प्रदूषक हैं।
स्रोत:
- आसमान में बिजली चमकने के कारण उत्पन्न अत्यधिक ताप पर N2 तथा O2 के संयोग से NO का बनना।
- ताप विद्युत् केन्द्रों में तथा ऑटोमोबाइल इंजन में अत्यधिक ताप के कारण NO का बनना।
- विभिन्न उद्योग में NO तथा ऑक्साइड का बनना।
- प्राकृतिक तथा जैविक क्रियाओं द्वारा NO का बनना।
N2 + O2 → 2NO
2NO + O2 → 2NO2
2NO + N2 → 2N2O
दुष्परिणाम:
- CO की तरह NO भी हीमोग्लोबिन की क्षमता को घटाता है।
- सूर्य प्रकाश में NO2 विघटित होकर परमाणवीय ऑक्सीजन देता है जो खतरनाक स्मॉग तैयार करता है।
- प्रकाश संश्लेषण की दर को घटाता है।
- श्वसन संबंधी रोगों को बढ़ाता है।
- रंजकों की चमक को नष्ट कर देता है।
नियंत्रण:
- विभिन्न प्रकार के ईंधनों को जलाने के लिये कुछ कम वायु रखी जाये तो NO के निर्माण की साम्यावस्था पीछे जायेगी जिससे NO का उत्पादन कम होगा।
- नाइट्रोजन के ऑक्साइड अम्लीय होते हैं इसलिये क्षारीय विलयनों द्वारा दूर किया जा सकता है।
- नाइट्रोजन के ऑक्साइड को उत्प्रेरकों द्वारा N2 या NH3 में अपचयन किया जा सकता है।
सल्फर के ऑक्साइड:
SO2 प्राथमिक प्रदूषक है तथा SO3 व H2SO4 द्वितीयक प्रदूषक हैं।
स्रोत:
- ज्वालामुखी से निकलने वाली गैसों में SO2 की पर्याप्त मात्रा होती है।
- कई जीवाणु में जैविक क्रियाओं के दौरान H2S गैस मुक्त होती है जो वायु द्वारा ऑक्सीकृत होकर SO2 देती है।
- कई धातुओं के अयस्क भी सल्फाइड तथा पाईराइटीज के रूप में पाये जाते हैं। धातुओं के निष्कर्षण में SO2 गैस निकलती है।
2ZnS + 3O2 → 2ZnO + 2SO2
2PbS + 3O2 → 2PbO + 2SO2 - जीवाश्म ईंधन जैसे-कोल तथा पेट्रोलियम में भी सल्फर होता है, जो दहन के पश्चात् SO2 बनाती है।
S + O2 → SO2
नियंत्रण:
- कारखानों से निकलने वाली SO2 युक्त गैसों के चूर्ण से उपचारित किया जाता है। जिससे यह CaSO4, में बदल जाती है।
CaO + \(\frac{1}{2}\)O2 + SO2 → CasO4 - बुझा हुआ चूना Ca(OH)2 (लाइम स्लेटी) के द्वारा SO2 दूर कर सकते हैं।
Ca(OH)2 + SO2 → CaSO3 + H2O - NaOH के द्वारा भी SO2 दूर कर सकते हैं।
2NaOH + SO2 → Na2SO3 + H2O
![]()
प्रश्न 5.
कुछ समय पूर्व, अंटार्कटिका के ऊपर ध्रुवीय समतापमण्डलीय बादल बने थे। ये क्यों बने थे? जब ये बादल सूर्य की गर्मी से विखंडित होते हैं, तो क्या होता है?
उत्तर:
गर्मी के मौसम में नाइट्रोजन डाइऑक्साइड तथा मेथेन, क्लोरीन मोनोऑक्साइड तथा क्लोरीन परमाणुओं के साथ अभिक्रिया करके क्लोरीन सिंक बनाते हैं, जो ओजोन क्षय को काफी हद तक रोकता हैं।
सर्दी के मौसम में, अंटार्कटिका के ऊपर विशेष प्रकार के बादल, ध्रुवीय समतापमण्डलीय बादल बनते हैं। ये ध्रुवीय समतापीमण्डलीय बादल एक प्रकार के सतह प्रदान करते हैं। जिसमें क्लोरीन नाइट्रेट जलयोजित होकर हाइपोक्लोरस अम्ल बनाता है। यह हाइड्रोजन क्लोराइड के साथ क्रिया करके आण्विक क्लोरीन बनाता है।
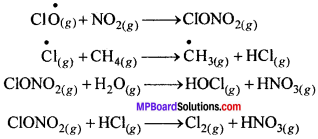
बसंत में जब अंटार्कटिका पर सूर्य का प्रकाश लौटता है, तो सूर्य की गर्मी बादलों को विखंडित कर देती है तथा HOCI तथा Cl, सूर्य के प्रकाश द्वारा अपघटित हो जाते हैं।
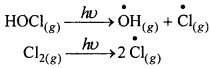
इस प्रकार उत्पन्न क्लोराइड मूलक ओजोन क्षय के लिए श्रृंखला अभिक्रिया प्रारंभ कर देते हैं।
प्रश्न 6.
पौधों से प्राप्त कम-से-कम सात औषधियों का वर्णन कीजिए।
उत्तर:
1. नीम-वानस्पतिक नाम – Azadirachta indica.
उपयोग:
- वातावरण को शुद्ध रखता है।
- नीम की पत्ती कीटाणुनाशक है।
- नीम का तेल घाव के उपचार में होता है।
2. आँवला-वानस्पतिक नाम – Emblica officinalis.
उपयोग:
- त्रिफला चूर्ण औषधि के निर्माण में।
- आँवला के बीज का पाउडर बाल धोने के काम आता है।
3. तुलसी-वानस्पतिक नाम – Ocimum sanctum.
उपयोग:
- इसे घरों तथा मंदिरों में लगाया जाता है ताकि वातावरण स्वच्छ रहे।
- इनकी पत्तियों का उपयोग बुखार तथा खाँसी के उपचार में किया जाता है।
- एन्टीसेप्टिक गुण होता है।
4.महआ-वानस्पतिक नाम – Madhuca indica.
उपयोग:
- पत्तियों, बीजों तथा फूलों का उपयोग चर्म रोग के उपचार में किया जाता है।
- इसके फूल से शराब बनायी जाती है।
5. अरण्डी -वानस्पतिक नाम – Ricinus communis.
उपयोग:
- इसके तेल का उपयोग साबुन बनाने में, मोमबत्ती बनाने में।
- तेल का उपयोग औषधि में होता है।
6. हर्रा-वानस्पतिक नाम – Terminalia chebula.
उपयोग:
- चमड़े की रंगाई में होता है।
- औषधि के निर्माण में।
- फल के पाउडर का उपयोग दमा के इलाज में उपयोगी है।
7.खस-वानस्पतिक नाम – Veliveraziga nicucles.
उपयोग:
- तेल का उपयोग सौंदर्य प्रसाधन, इत्र तथा पेय बनाने में।
- एक कीट प्रतिकर्षी है।
- इनका उपयोग गठिया, वात, कमरदर्द तथा मोच के उपचार में होता है।
- यह उत्तेजक तथा प्रशीतक
प्रश्न 7.
आप निम्नलिखित के लिए हरित रसायन का प्रयोग किस प्रकार करेंगे –
- प्रकाश रासायनिक धूम-कोहरे के नियंत्रण में।
- कपड़ों की निर्जल धुलाई में हैलोजनीकृत विलायक तथा विरंजन में क्लोरीन के उपयोग से बचाव में।
- संश्लेषित अपमार्जकों के उपयोग को कम करने में।
- पेट्रोल तथा डीजल के उपयोग को कम करने में।
उत्तर:
- कुछ पौधे जैसे-पाइनस, जुनीपेरस, क्वेरकस, पायरस तथा विटिस, नाइट्रोजन ऑक्साइड (NO) का उपापचय कर सकते हैं। अत: इनके रोपण द्वारा प्रकाश रासायनिक धूम-कोहरे को नियंत्रित किया जा सकता है।
- कपड़ों की निर्जल धुलाई में हैलोजनीकृत विलायक के स्थान पर उचित अपमार्जक के साथ द्रवीकृत CO2 तथा कपड़ों, कागज के विरंजन में Cl2 के स्थान पर H2O2 के प्रयोग के अच्छे परिणाम आते हैं तथा जल का बहुत कम उपयोग होता है।
- साबुन 100% जैवनिम्नीकृत होते हैं अतः इनका प्रयोग अपमार्जकों के स्थान पर किया जा सकता है। आजकल जैव-निम्नीकृत अपमार्जक भी उपलब्ध हैं। अतः अजैवनिम्नीकृत कठोर अपमार्जक के स्थान पर इनका प्रयोग किया जाना चाहिए।
- C.N.G. (संपीडित प्राकृतिक गैस) का प्रयोग किया जाना चाहिए क्योंकि यह बहुत कम प्रदूषण करती है। साथ ही, विद्युतीय वाहन के प्रयोग द्वारा भी पेट्रोल तथा डीजल की खपत कम की जा सकती है।
प्रश्न 8.
लंदन (सामान्य) कोहरा व प्रकाश रासायनिक कोहरा में अंतर स्पष्ट कीजिए।
उत्तर:
लंदन (सामान्य) धूम कोहरा व प्रकाश रासायनिक धूम कोहरा में अंतर –
सामान्य धूम कोहरा
- इस प्रकार का धूम कोहरा प्रथम बार 1952 में में देखा गया था।
- यह SO2 आर्द्रता का H2SO4 से क्रिया करने पर बनता है, जो कणिकीय पदार्थ के रूप में जमा होता है।
- यह धुआँ और कोहरे से बनता है।
- वह सुबह के समय सर्दी के मौसम में बनता है।
- यह फेफड़ों में रोग उत्पन्न करता है।
- यह अपचायक गुणधर्म दर्शाता है।
प्रकाश रासायनिक धूम कोहरा
- यह धूम कोहरा प्रथम बार लॉस ऐंजिलीस में 19501 लंदन में देखा गया था।
- यह प्रकाश रासायनिक क्रिया से जो NO2 व हाइड्रोकार्बन के बीच संपन्न होती है।
- यह धुआँ या कोहरा नहीं रखता है।
- यह गर्मीयों के दिन के मध्यांतर में बनता है।
- इससे आँख में जलन उत्पन्न होती है।
- यह ऑक्सीकारक गुणधर्म दर्शाता है।
![]()
प्रश्न 9.
(a) मृदा प्रदूषण के प्रमुख स्रोत क्या हैं?
(b) पर्यावरण प्रदूषण को कैसे नियंत्रित किया जा सकता है?
उत्तर:
(a) मृदा प्रदूषण के प्रमुख कारण निम्न हैं –
- औद्योगिक अवशेष
- कीटनाशी, पीड़कनाशी तथा शाकनाशी
- उर्वरक, D.D.T.., B.H.C., NaClO3, Na3AsO3, आदि,
- रेडियोधर्मी पदार्थ।
(b) पर्यावरणीय प्रदूषकों में घर में उत्पन्न कूड़ा-करकट, औद्योगिक अवशेष आदि हैं। जिन्हें निम्न विधियों द्वारा नियंत्रित किया जा सकता है –
- पुनः चक्रण करके, प्रयोग किया गया काँच, प्लास्टिक, लोहा, पॉलीथीन, कागज का पुनः चक्रण किया जावे।
- दहन से।
- सीवेज व्यवस्था से।
- अपशिष्टों का उचित प्रबंधन करके।
- हरित रसायन का प्रयोग करके।
- जन-जागृति द्वारा।
- पेड़-पौधों को अधिक मात्रा में लगाकर।
- नियंत्रक यंत्रों तथा युक्तियों को अपनाकर।
- प्रदूषक निकालने वाले यंत्रों को उच्च उन्नतांश तक लगाकर (ऊँची चिमनी के प्रयोग द्वारा)।