Students get through the MP Board Class 11th Chemistry Important Questions Chapter 14 Environmental Chemistry which are most likely to be asked in the exam.
MP Board Class 11th Chemistry Important Questions Chapter 14 Environmental Chemistry
Environmental Chemistry Class 11 Important Questions Very Short Answer Type
Question 1.
Define Environmental chemistry.
Answer:
Environmental chemistry is a branch of science which deals with the study of effects of chemicals on our surroundings (like origin, transport, reactions and effects and fates of chemical species etc.).
Question 2.
Explain Tropospheric pollution.
Answer:
The tropospheric pollution occurs because of the presence of undesirable gaseous and solid particles in the air. In the troposphere mainly the following gaseous and particulate pollutants are present.
- Gaseous air pollutants are oxides of sulphur, nitrogen and carbon dioxide, hydrogen sulphide, ozone and other oxidants.
- Particulate pollutants are dust, fumes, mist, spray, smoke etc.
Question 3.
Make a list of the gases responsible for greenhouse effect.
Answer:
For greenhouse effect mainly CO2 gas is responsible. Apart from CO2, other greenhouse gases are methane, water vapour, nitrous oxide, chlorofluorocarbon (CFC) and ozone.
Question 4.
What do you mean by Biochemical oxygen demand BOD?
Answer:
The amount of oxygen required by bacteria to break down the organic matter present in a certain volume of a sample of water is called Biochemical oxygen demand (BOD). Amount of BOD in the water is a measure of the amount of organic material in the water, in terms of how much oxygen will be required to break it down biologically. For pure and clean water BOD value is less than 5 ppm.
![]()
Question 5.
Due to greenhouse effect, earth’s temperature is increasing. Which substances are responsible for greenhouse effect?
Answer:
Greenhouse gases like carbon monoxide, methane, nitrous oxide, ozone and chlorofluorocarbons (CFCs) are responsible for greenhouse effect. These gases absorb the radiations passing through the earth’s surface and increase the earth’s temperature.
Question 6.
Ozone is a poisonous and a strong oxidizing reagent still its presence in the upper part of stratosphere is very important. What will happen if ozone is completely removed from this region?
Answer:
Ozone prevents the harmful ultraviolet rays of the sun to reach the earth’s surface due to which human life is protected by the harmful effects of these ultraviolet rays.
If ozone is completely removed from the upper region of stratosphere, then ultraviolet rays will reach the earth and cause various diseases like skin cancer etc.
Question 7.
What are the sources of oxygen dissolved in air?
Answer:
The sources of oxygen dissolved in water are :
- Photosynthesis,
- Natural Aeration,
- Mechanical Aeration.
Question 8.
Oxygen dissolved in water is very important for aquatic life. Which factor is responsible for the reduction of dissolved oxygen in water?
Answer:
For the reduction of dissolved oxygen in water, discharge of phosphate and nitrate fertilizers, detergents, animal excreta, food, paper and pulp industry, organic waste from industries are responsible. Micro-organisms which oxidize organic substances also use dissolved oxygen. Along with this, photosynthesis stops at night but aquatic plants and animals continuously breathe due to which deficiency of dissolved oxygen in water is caused.
![]()
Question 9.
What are Biodegradable and non-biodegradable pollutants?
Answer:
Biodegradable pollutants are substances which are decomposed by organisms.
Example: Excreta, cow dung, fruits and vegetable peels.
Non-biodegradable pollutants are substances which are not decomposed by organisms.
Example: Mercury, Lead, DDT, glass, plastic etc.
Question 10.
What is pollution?
Answer:
In a balanced environment, every component is present in a definite amount and ratio. Sometimes, in the environment amount of one or more component either increases than required or harmful components enter into the environment which are harmful for living organism. This is known as pollution.
Question 11.
What are pollutants?
Answer:
Such substances whose amount increases up to a certain limit in the atmosphere due to which it becomes harmful for plant and animal kingdom are called pollutants. They are as follows:
CO, CO2, NO, N2, S2 etc.
Question 12.
What are contaminants?
Answer:
Some substances do not exist in the environment but are made in artificial form by man. These substances pollute the environment without getting destroyed. They are called contaminants.
Question 13.
Write the names of gases or chemical responsible for the depletion of ozone layer.
Answer:
Nitric oxide, atomic oxygen and chlorofluorocarbon present in the atmosphere is responsible for the depletion of ozone layer.
Question 14.
What are greenhouse gases?
Answer:
CO2, ozone and water vapour are known as greenhouse gases. They possess the property of absorbing infrared radiations. Thus, they are known as greenhouse gases.
Question 15.
What is polluted air?
Answer:
If air contains such substances which are harmful for health of organisms, then such air is called polluted air. Polluted air mainly contains suspended molecules of CO2, SO2, SO3, CO.
Question 16.
Why is depletion of ozone observed Antarctica?
Answer:
Because in the other part of stratosphere, free chlorine gets converted to chlorine molecules, whereas at Antarctica compounds produced get surrounded by chlorine free radical which cause depletion of ozone.
![]()
Question 17.
What is the utility of BOD measurement in a water sample?
Answer:
BOD is measure of pollution caused by organic biodegradable substances present in a given sample of water. Lower value of BOD indicates that less amount of organic waste is present in water.
Question 18.
Oxidation of sulphur dioxide to sulphur trioxide is a slow change in the absence of a catalyst but in presence of atmosphere it oxidizes readily. Why is it so? Also give chemical equation for the conversion of SO2 to SO3.
Answer:
In the oxidation of SO2 to SO3, particulate matter present in polluted air acts as a catalyst.
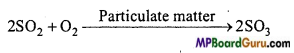
Question 19.
How is ozone produced in stratosphere?
Answer:
In stratosphere, ozone is produced by the action of ultraviolet radiations on dioxygen (O2) molecule. Ultraviolet rays decompose oxygen molecule to oxygen atom. Oxygen atom combine with oxygen molecule to form ozone molecule.

Question 20.
What is chlorosis?
Answer:
In plants, chlorophyll is formed slowly This is due to presence of SO2. This pollution is called chlorosis.
Question 21.
What is Metathesis?
Answer:
Metathesis is the name of that science, in which utility of scientific methods for a common man is studied.
Question 22.
What are Primary and Secondary Pollutants?
Answer:
Primary Air Pollutants: Air pollutants which directly pollute the air by natural or human activities are called primary pollutants. Like CO, NO, CO2, SO2.
Secondary Air Pollutants: Such air pollutants which are prepared by the chemical reactions between pollutants or by the reaction of gases and pollutants in the atmosphere are called secondary pollutants.
Like: SO3+H2O → H2SO4.
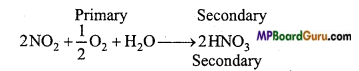
Question 23.
What is photochemical smog?
Answer:
Mixture of smoke and fog is known as smog. When higher concentration of oxidizing agents are present in this smog, it is called photochemical smog. Smoke is an air pollutant, in which various solid particles are suspended in the atmosphere due to incomplete combustion of fuel substances.
Question 24.
What is Acid rain?
Answer:
Due to air pollution, concentration of various gases like CO2, SO2, SO3, NO2 etc. increases in the atmosphere. These gases dissolve in raindrops according to their solubilities and form acid. These drops fall as rain and are known as acid rain.
CO2+H2O → H2CO3
SO3+H2O → H2SO4.
Question 25.
When is CO2 known as pollutant?
Answer:
Normal amount of CO2 in the atmosphere is never harmful, but plants prepare their food by it. But, when due to various activity its concentration becomes more than required, then it disturbs the natural balance and become harmful.
![]()
Question 26.
Carbon dioxide is considered to be more responsible for greenhouse effect. Why?
Answer:
Water vapour is found to be very close to the earth, and ozone is found at the upper part of the atmosphere. Compared to this CO2 is evenly found in all places of the atmosphere. Therefore, for greenhouse effect, CO2 is found to be more responsible because CO2 possess the tendency to absorb the infrared radiations of the sun and produce greenhouse effect.
Question 27.
Why is acid rain considered as a threat to Tajmahal?
Answer:
Tajmahal is made of marbles. The acid rain contains H2S04 which attacks the marble (CaCO3).
CaCO3(s) + H2SO4(aq) → CaSO4(aq) + CO2+ H2O(l)
As a result, the marble becomes pitted and weakened mechanically. Therefore, the monument is being slowly eaten away and marble is getting discoloured and lustreless.
Question 28.
A person uses water supplied by municipality. Due to shortage of water, he started using underground water. He felt laxative effect. What could be the cause?
Answer:
Laxative effect can be felt only when the concentration of sulphate present in water is greater than 500 ppm otherwise at moderate concentration sulphate is harmless.
Question 29.
What do you understand by Green chemistry? How is it helpful to control environmental pollution?
Answer:
Green chemistry is a technique to utilize the known knowledge and principles of chemistry and other sciences by which harmful effects of environment can be reduced. Green chemistry is a process of production which can bring minimum pollution or harm to the environment. Co-products produced in a process if not removed in a beneficial way increase pollution in the environment. Such processes are not only harmful for environmental view but are also costly. Along with modified work, use of present knowledge to reduce chemical harm is the basis of Green chemistry.
Question 30.
Why is carbon monoxide more dangerous than carbon dioxide? Explain.
Answer:
Carbon monoxide is a poisonous gas. It combines with haemoglobin present in blood to form carboxyhaemoglobin. It is 300 times more stable than oxyhaemoglobin. When the percentage of carboxyhaemoglobin in the blood becomes 3-4% then ability of the blood to carry oxygen becomes very less.
Due to minimum oxygen, headache, weak eyesight, reduction in awareness and judgement, cardiovascular diseases etc. are produced. Carbon dioxide gas does not combine with haemoglobin. Therefore, it is a less harmful pollutant. Increase in its amount in the environment leads to global warning (increase in earth’s temperature).
Question 31.
What is Fly ash pollutant?
Answer:
Fly ash are small ash particles formed by the combustion of fossil fuels which are carried into the air by gases produced during combustion.
Question 32.
What is contaminated water? Write name of diseases caused due to contaminated water.
Answer:
Water in which due to pollution various harmful products of living beings and micro-organisms are present is called contaminated water. Diseases caused due to contaminated water are :
- Cholera,
- Jaundice,
- Diarrhoea,
- Skin disease.
Question 33.
What do you understand by Global warming?
Answer:
Increase in earth’s temperature, due to absorption of sun’s energy by various gases like CO2, methane, chlorofluorocarbon etc. present in large amount in the atmosphere is called Global warming.
![]()
Question 34.
What is Ionosphere?
Answer:
A thick layer about 40 km at a height of about 50 to 90 km from the earth’s surface is called Mesosphere. All the gases in this region exist in ionized state, therefore it is called Ionosphere.
O2 + O+ → O2 + O
O+ + N2 → NO+ + N
N2+O2 → N2 + O2.
Environmental Chemistry Class 11 Important Questions Short Answer Type
Question 1.
How is the poisonous effect of CO produced on man and animals?
Answer:
CO has poisonous effect on man and plants.
(i) It combines with haemoglobin of the blood more strongly than oxygen. CO + Hb → CO – Hb (Carboxy haemoglobin).
As a result of this amount of haemoglobin available in the blood for the transport of oxygen to the body cells decreases. The normal metabolism is thus, impaired due to less O2 level. This will cause suffocation and will ultimately lead to death. Carbon monoxide if present in air can cause mental impairment, respiratory problems, muscular weakness and dizziness.
(ii) A high concentration of CO (100 PPM or more) will harmfully affect the plants causing leaf drop; reduction of leaf size and premature aging etc.
Question 2.
Write the harmful effects of sulphur dioxide.
Answer:
Harmful effects of SO2 :
- SO2 harms the respiratory tube and lungs due to which various lung diseases, cancer etc. can be caused.
- It stops the growth of plants. Due to this, change of green leaves to yellow is called chlorosis.
- Acid rain due to SO2 harms the monuments and stones.
- SO2 gas enhances corrosion.
Question 3.
Write harmful effects of nitrogen oxide.
Answer:
Harmful Effects of Nitrogen oxide :
- Presence of gases like NO, NO2 etc. more than required cause irritation in eyes.
- Its excess amount may lead to lung and heart diseases.
- Oxides of nitrogen enhance corrosion in metals.
- Reduce rate of photosynthesis.
- Reduce the brightness of dyes.
Question 4.
A farmer was using pesticides on his farm. He used the produce of his farm as food for rearing fishes. He was told that fishes were not fit for human consumption because large amount of pesticides had accumulated in the tissues of fishes. Explain how did it happen?
Answer:
Pesticides transfer from the crops to fish as their food. This way, pesticides through the food chain reach the higher level from the lower level. With time, concentration of pesticides in fish increased to such a level that serious metabolic and body active states were produced. That is why, fishes were not suitable as food for man.
Question 5.
For dry cleaning, in place of tetrachloroethene along with suitable deter-gent liquified carbon dioxide is an alternative. Which loss of the environment can be stopped by the use of tetrachloroethene? Is the use of liquified carbon dioxide with detergent a completely prevention from the view of pollution?
Answer:
- Tetrachloroethene (Cl2C = CCl2) is a possible cause of cancer, it also pollutes ground water. Its harmful effects can be controlled by the use of liquified carbon dioxide and suitable detergent.
- Use of liquified CO2 with suitable detergent is not completely preventive because maximum detergents are non-biodegradable and they also pollute the water. Along with this, liquified CO2 enter the environment at the end and increase green house effect.
Question 6.
What are the ill effects of greenhouse effect?
Answer:
Main cause of greenhouse effect is the presence of large amount of CO2 in the atmosphere which absorbs the infrared radiations and increase the temperature of the environment. This way, due to increase in temperature of the environment weather will change. Sun rays gives birth to serious disease like cancer.
Some places will suffer from drought, hot air will flow in some places, some places will have strong storm and somewhere floods may come. Huge icecaps of Arctica and Antarctica will melt, by which sea level will rise and places near sea shores will submerge.
![]()
Question 7.
What is smog? Explain its mechanism with its types.
Answer:
Mixture of smoke and fog is known as smog.
Types of Smog:
- Classical smog: SO2 gas mixed with fog combines with smoke to form classical smog.
- Photochemical smog: In a Sunny place, primary air pollutant form secondary pollutant by photochemical reaction. They are oxidizing in nature. Where pollutant gas is more, wind does not blow, there with smoke, smog is formed.
Mechanism :
1. If automobile fuel is used before sunrise, then mainly CO and NO gas are released. With contact of air it forms NO2 gas.
2NO + O2 → 2NO2
2.By the effect of ultraviolet rays active oxygen is released by NO2.
NO2 → NO + O
3. This active oxygen converts the hydrocarbon released from the fuel to free radical.
4. These organic free radicals form undesirable compounds by chain reactions and these pollutants form smog.
Question 8.
Discuss the causes of water pollution.
Answer:
Following are the causes of water pollution :
- Waste products from big factories are dumped in sea, rivers or lakes.
- Various fertilizers, manure and insecticides are used in fields.
- All these products mix with the soil of fields, dissolve in rainwater and fall in rivers and pools.
- Atomic electric centre pollute the water.
- Due to increase in population, colonies are formed at the river banks, and waste from the houses pollute the water.
- In rural areas, people take bath, wash clothes in the rivers which pollute water.
Question 9.
How is synthetic greenhouse prepared?
Answer:
Synthetic greenhouse: In nature, coating of CO2 is forming greenhouse, but synthetic greenhouse can be synthesized by studying its mechanism.
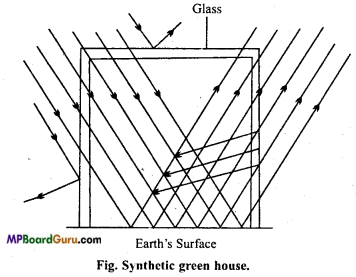
Actually the transparent glass roof and wall of the glasshouse allow sun rays to pass through and strike the surface of the house. The reflected radiation is of longer wavelength than the incident radiation. A significant portion of reflected radiation absorbed by glass. As radiation of longer wavelength (Infrared radiation) generates heat, this causes rise in tem¬perature inside the glasshouse.
Question 10.
You have dug pits in your agricultural area or garden for producing compost. Discuss the process of production of best compost in the light of bad odour, Hies and recycling of waste.
Answer:
For producing compost, pits should be dug in suitable place by which we can be prevented from bad odours and flies. Biodegradable domestic waste like tea leaves, fruits and vegetable waste etc. should be put in the compost pit and should be covered by thin layer of sand. After some time, these waste materials, by the effect of heat and bacteria get converted to compost.
Compost pits should be covered by sand by which flies cannot enter and bad odour is minimum.
Non-biodegradable waste like plastic, glass, metal shavings, polythene bags are sent for recycling. By recycling useful products are obtained from waste products.
![]()
Question 11.
How are domestic waste used as manure?
Answer:
Domestic wastes are collected in small containers and sent to disposal area. Here biodegradable waste and non-biodegradable waste are separated from the garbage. Biodegradable waste like vegetables and fruit waste, animal waste etc. are in open ground beneath the soil where it degrades and after some days get converted to manure.
Question 12.
Write the ill effects of water pollution and how is it controlled.
Answer:
III effects of water pollution:
- By the use of polluted water, cholera, jaundice, typhoid like contaminated diseases are spread.
- Due to discharged excreta, amounf of oxygen dissolved in water decreases due to which aquatic organisms die.
- Due to soap, detergent waste, polluted water becomes a danger for the life of fish.
Control:
- Discharged excreta should not be thrown in rivers, pools etc. by chemical reaction should be converted to manure.
- Atomic identification below the sea should be banned.
- By the process of recycling cow dung and excreta should be converted to useful material.
Question 13.
What is soil pollution? How can soil pollution be controlled?
Answer:
Soil Pollution: Any undesirable change in physical, chemical or biological properties of soil which has harmful effect on human beings and other organisms or the natural character and utility of soil or earth is destroyed is called soil pollution.
Control:
- Industrial waste should be treated in the factories only and then dumped on earth.
- Use of fertilizers, insecticides and fungicidal medicines should be minimized.
- Cutting of trees should be stopped to prevent soil erosion and destruction of the layer of earth.
Question 14.
Write the ill effects of depletion of ozone layer.
Answer:
Following is the ill effects of ozone layer :
- Entrance of ultraviolet rays directly on earth without obstruction can lead to various diseases like cancer.
- Ultraviolet rays decrease the immunity of body.
- CFC increases greenhouse effect, as a result temperature of earth rises.
Control:
- Production of CFC should be reduced.
- Search the option of CFC.
Question 15.
Explain the mechanism of depletion of ozone layer.
Answer:
The mechanism of depletion of ozone layer are the following :
1. Depletion by Nitrogen oxide: Some amount of N2O is present in the stratosphere. It combines with atomic oxygen to form NO.
N2O+O → 2NO
NO gas combines with O3 to form NO2
NO + O3 → NO2 + O2.
NO2 breaks by atomic oxygen to form NO again
NO2+O → N0 + O2
This cycle of NO is the cause of depletion of O3.
2. Depletion by C.F.C.: It produces free radical by photochemical decomposition.
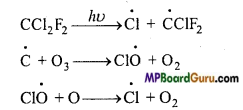
Formation of free radicals is continuously depleting ozone layer.
Environmental Chemistry Class 11 Important Questions Long Answer Type
Question 1.
A large number of fish are suddenly found floating dead on a lake. There is no evidence of toxic dumping but you find an abundance of phytoplankton. Suggest a reason for fish kill.
Answer:
Large amount of phosphate and nitrate salts increases the number of phytoplanktons in water. These phytoplanktons use the dissolved oxygen in water to such a large extent, that it is not available in sufficient amount for the respiration of other organisms. Along with it excessive population of bacteria start the decomposition of organic substance in water like leaves, grass etc. It uses the dissolved oxygen in water. Micro-organisms (like algae) are in such a larger amount that they cover the water level as a sheet and prevents the penetration of sunlight in the water level as a result photosynthesis is obstructed. By all these processes, amount of dissolved oxygen decreases 6 ppm. Thus, growth of fish is stopped and they do not remain alive.
Question 2.
What would have happened, if there would have been no greenhouse gases in the atmosphere?
Answer:
Carbon dioxide, methane, water vapour, CFCs and ozone are green house gases. These gases absorb the developed heat by the objects situated near the earth’s surface and heat it up. This is known as natural green house effect because it controls temperature and makes life better on earth. If there would have been no green gases in the atmosphere, then there would have been no vegetation or life present on earth (because earth would have converted to a cool house).
Question 3.
Into how many parts is the atmosphere divided. Explain the chemical reactions occuring in every region.
Answer:
Atmosphere is divided into four parts :
- Troposphere,
- Stratosphere,
- Mesosphere,
- Thermosphere.
1.Troposphere: The region upto a height of 8 to 12 km from the earth’s surface is called Troposphere. Its main components are N2, O2, CO2 and H2O. Fossil fuels bum in oxygen to form CO2.
CH4 +2O2 → CO2 +2HzO
In the troposphere, compounds are decomposed by various bacteria.
Bacteria compound + O2 → CO2 + H2O
Photosynthesis also occur in this layer.
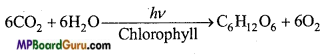
2. Stratosphere: About 10 to 50 km, layer of 40 km thickness is known as Stratosphere.
(a) Photochemical decomposition of oxygen.
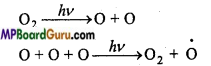
Excited oxygen molecule 02 liberate radiation of 636, 630, 558nm wavelength in visible region. This is known as airglow.
![]()
Oxygen molecule gets excited and lose electron by which positively charged oxygen ion is formed.
O2 +O → O3
O3 molecule again decomposes by ultraviolet rays.
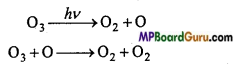
O3 + O → O2 + O2
To provide stability to ozone, others like N2, O2 are required which absorb the extra energy and stop the decomposition of O3.
O + O2+M → O3 + M
Alternatively, if species like NO or OH are present in stratosphere, then they decompose o3.
O3 + OH → O2 + HOO
O3 + NO →NO2 + O2
NO2 +O → NO + O2
N2 molecule undergo self decomposition by radiation and combine with O2 to form NO.
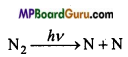
O2 + N → NO + O
3. Mesosphere: About 50 to 90 km height above the earth’s surface a 40 km thick layer is called Mesosphere.
O2 + O → O2 + O
O+ + N2 → NO+ + N
N2++O2 → N2 + O2
4. Thermosphere: This layer of atmosphere starts from a height of 90 km from the earth’s surface. This region till 500 km height is known as Thermosphere.
![]()
Question 4.
How is acid rain damaging statues and monuments in India?
Answer:
In India, statues and monuments are made up of marble i.e. CaCO3. The surrounding air mainly contain oxides of sulphur and nitrogen in large amount. The main reason is the presence of large number of industrial units and energy plants around it. Oxides of sulphur and nitrogen are acidic. SO2 and NO2 oxidize with water to form mineral acids which is the main source of acid rain.
2SO2(g) + O2(g) + 2H2O(l) → 2H2SO 4(aq)
4NO2(g) + O2(g) + 2H2O(l) → 4HNO3(aq)
This acid rain reacts with the marble of statues and monuments and destroy them.
CaCO3 + H2SO4 → CaSO4 + H2O + CO2
Question 5.
What is smog? How is classical smog different from photochemical smog?
Answer:
The word smog is derived from smoke and fog. This is the most common example of air pollution that occurs in many cities throughout the world. There are two types of smog.
- Classical smog: It occurs in cool humid climate. It is a mixture of smoke, fog and sulphur dioxide. Chemically it is a reducing mixture.
- Photochemical smog: Photochemical smog occurs in warm, dry and sunny climate. The main components of photochemical smog result from the action of sunlight on unsaturated hydrocarbons and nitrogen oxides produced by automobiles and factories. Chemically it is oxidizing smog as it contains high concentration of oxidizing agents.
Question 6.
How many types of particulate are there? Explain.
Answer:
Particulate: Finely divided solid or liquid particles suspended in air are the cause of pollution. Size of these particles can be from 0.02 Å to 50000Å. Our eyes can see particles of the size about 10000Å.
Various types of particles and their source :
1.Soot: Due to incomplete combustion of carbon-containing fuels minute particles of carbon spread in the environment. This type of combustion generally occur at all places because from wood, coal etc. used as domestic fuels carbon-containing fuels are used in every field.
2. Dust: Sources of dust are present at all places on the earth. These are released in the environment due to natural or human sources during volcanic eruption, storm, during certain industrial processes like crushing, grinding of limestone, cement as fly ash and silica dust. Apart from this, dust is also spread due to running of vehicles.
3. Metal particles: Particles of metals like lead, mercury, chromium, arsenic, zinc, iron, nickel, cadmium etc. spread in the environment by various types of metal treatment.
Though their quantity is very less, still they produce harmful effect even if present in minute quantity.
4. Metal oxide: Generates by combustion of fuels containing metallic compounds.
For example when coal containing iron pyrites is burnt, particulates of Fe3O4 are introduced into the air.
3FeS2+8O2 → Fe3O4 + 6SO2
5. Particles of metal salts and fly ash: Limestone present in coal is left in ash in the form of CaO and can fly with ash in dust. Minute drops of H2SO4 situated in the environment react with CaO to form particles of CaSO4. Ammonia present in air react with H2SO4 and form particles of (NH4)2SO4. The flying of ash with air in the form of minute particles producing pollution is called fly ash pollution.
6. Asbestos particles: Asbestos is used in the factories and industries in various forms. Therefore asbestos particles spread in the environment.
7. Mist: We know that water vapour condense into minute droplets in the atmosphere which appear as mist to us. Similarly acids like H2SO4 and HNO3 formed by various activities in the atmosphere also produce mist in the form of minute particles and spread pollution.
8. PAH and TEL: PAH means Polycyclic Aromatic hydrocarbon. TEL means Tetra Ethyl Lead. Due to incomplete combustion of fossil fuels and petroleum, PAH pollutes the environment in the form of minute particles. Tetraethyl lead, Pb (C2H5)4 is added to gasoline as antiknock agent.
During combustion in the engine, it is oxidized to PbO which deposits in different parts of the engine and may cause damage. To avoid this damage, Pb(C2H5)4 is mixed with dichloroethane and dibromo ethane which converts PbO into PbCl2 and PbBr2 which are volatile and thus come along with the exhaust gases and introduce into the atmosphere.
Pb(C2H5)4 + O2 + C2H4C12 + C2H4Br2 → CO2 + H2O + PbCl2 + PbBr2
Thus, engine is protected but air gets polluted.
![]()
Question 7.
What is Acid rain. Explain in detail its effect on the environment.
Answer:
All pollutant gases spread as pollutant particles in the form of smoke by the burning of fossil fuels and other fuels. Due to high temperature of industries and other engines, oxides of nitrogen spread in the atmosphere by the combination of N2 and O2. These gases mix in raindrops form acid and then fall on the earth as acid raindrops. This is called acid rain.
CO2 + H2O → H2CO3
SO2 + H2O → H2SO3
Effects of Acid rain :
- Acid rain neutralizes the base present in the soil, as a result of which soil becomes acidic. This affects the bacteria, plants and nitrogen fixation.
- Due to acid rain, water of rivers, lakes become acidic which has harmful effect on organisms.
- Acid rain leads to depleting forest resources and vegetation.
- Statues and monuments are losing their brightness because the metals present in them get corroded. Due to acid rain marble of Tajmahal is depleting.
- Acid rain harms leaves of aquatic plants and reduces the rate of photosynthesis.
- Acid rain has harmful effect on skin, lungs and neck.
Control of Acid rain :
- Rivers and lakes cannot be prevented from acid rain but by mixing lime, its effect can be reduced.
- By pouring lime in the fields, effect of acid rain can be reduced.
- By using coal with low content of sulphur, unleaded petrol process of combustion of fuel in plants can be controlled and effect of acid rain can be destroyed.
- Measures to control the effect of gases causing acid pollution and converting unburnt hydrocarbons to ineffective compound are being tried.
Question 8.
Explain the process of pollution of oxides of sulphur and nitrogen. How can they be controlled?
Answer:
Oxide of sulphur (SOx): Among the oxides of sulphur, SO2 is the main pollutant. It gets partially oxidised to SO3 by air. SO3 forms H2SO4 in presence of moisture this way SO2 is a primary pollutant and SO3 and H2SO4 is a secondary pollutant.
Process of pollution : (i) Fossil fuels and petroleum contain 0-3% to 3% of S which form S02 on burning.
S+O2 → SO2
(ii) SO2 gas is released by industrial extraction of metals.
2FeS2 + 5O2 → 2FeO + 4SO2
2PbS + 3O2 → 2PbO + 2SO2
2ZnS + 3O2 → 2Zn0 + 2SO2
(iii) During biological activities of various bacteria H2S gas is released which oxidizes by air to form SO2.
H2S+\(\frac{3}{2}\) O2 → SO2+H2O
(iii) Volcanic gases contain sufficient amount of SO2
Control:
- To control SO2, impurity of S should be removed from fuels.
- To remove sulphur from the engine by physical methods it is washed properly by making powder.
- In chemical methods, fossil fuels are carbonated.
- Liquation and vaporization are other methods of sulphur control.
- Environmental SO2 can be controlled by scrubbing.
Oxides of nitrogen (NOx): Nitric oxide is primary pollutant whereas N2O, NO2 or other oxides are secondary pollutant which are formed in the environment by various chemical activities.
Source:
- Due to high temperature produced by lightning in the atmosphere N2 and O2 combine to form NO.
- Production of NO due to high temperature in automobile engine.
- Formation of NO and other oxides in various industries.
- Formation of NO by natural and biological activities and due to fire in jungle.
Control:
- In the combustion of fuel, on keeping the amount of airless, the equilibrium
for the manufacture of NO can be shifted to backward which can control the formation of NO.
N2 + O2 ⇌ double arow 2NO - Oxides of nitrogen released from automobile engines can be reduced by catalyst like Pt to N3 or NH3.
- Oxides of N2 are acidic, therefore can be removed by basic solution.
- Scrubbing of nitrogen oxides can be done by acids.
Question 9.
Have you seen soil pollution in the nearby area? What measures will you take to control soil pollution ?
Answer:
Yes, it can be controlled by the following methods :
1. insecticides and pesticides which are used to protect our crops spread soil pollution. Therefore its use should be limited.
2. After the second world war, DDT was used in agriculture to control insects, pests, weeds and various crop diseases. Due to its adverse effect, its use was banned in India. Pesticides like Aldrin and Dieldrin are actually toxic. These are insoluble and non-biodegradable in water due to which they cause serious metabolic and physiological disorders.
Nowadays organophosphates and carbamates are used as pesticides. They are more biodegradable compounds but they are serious toxins and are harmful to human beings. Thus, fertilizers, detergents, pesticides, polymers like chemicals are used only under extreme necessity.
3. Biodegradable domestic waste should be dumped in pits.
4. Non-biodegradable waste should be recycled.
5. Use of polythene should be avoided.
6. Domestic waste, biological waste and chemical waste should be burnt. As a result of incineration volume of waste products is reduced.
Question 10.
Sometime ago formation of polar stratosphere clouds was reported over Antarctica. Why these formed? What happens when such clouds break up by warmth of sunlight?
Answer:
In summer season, nitrogen dioxide and methane react with chlorine monoxide and chlorine atoms and form chlorine sink which prevent ozone depletion to a large extent.
In winter season, special type of clouds, polar stratosphere clouds are formed over Antarctica. These clouds provide a type of surface by which chlorine nitrate hydrolysis to form hypochlorous acid. It reacts with hydrogen chloride to form molecular chlorine.
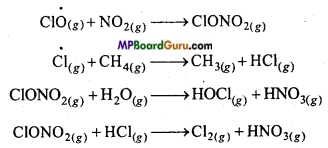
In spring, when sunlight return to Antarctica, heat of sun disperse the clouds and HOCl and Cl2 decompose by sunlight.
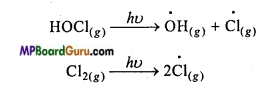
This way, the chloride radical produced start the chain reaction for the depletion of ozone.
Question 11.
Describe at least 7 medicines obtained from plants.
Answer:
1. Neem: It is the plant of family Malice.
Uses :
- It keeps the environment pure,
- Neem leaves are insecticides,
- Neem oil is used in the treatment of wound.
2. Amla: Botanical name: Emblica Officinalis.
Uses :
- Used to prepare Triphala powder,
- Powder of seeds of Amla are used for washing hairs.
3. Tulsi: Botanical name: Ocimum sanctum.
Uses :
- It is grown in houses and temples to keep the environment clean,
- Its leaves are used for the treatment of fever and cough,
- It has antiseptic property.
4. Mahua: Botanical name: Madhuca indica.
Uses :
- Leaves, seeds and flowers are used for the treatment of skin disease,
- Its flowers are used to prepare wine.
5. Arandi: Botanical name: Ricinus communis.
Uses :
- Its oil is used to prepare soap, candles etc.
- Oil is used as medicine.
6. Harra : Botanical name : Terminalia chebula.
Uses :
- In dyeing leather,
- In the manufacture of medicines,
- Powder of fruit is used for the treatment of asthma.
7. Khus: Botanical name: Velivera ziga includes.
Uses :
- Oil is used in cosmetics, perfume and making beverage,
- It is insect repellant.
- It is used in the treatment of arthritis, backache, sprain etc.
Question 12.
How will you use Green chemistry for the following :
1. To control photochemical smog.
2. To avoid use of halogenated solvents in dry-cleaning and that of chlorine in bleaching.
3. To reduce use of synthetic detergents.
4. To reduce the consumption of petrol and diesel.
Answer:
1. Some plants like Pinus, Pyrus can metabolise nitric oxide (NO). Thus, their growth can control photochemical smog.
2. In dry-cleaning, use of halogenated solvent is replaced by a process, where liquified CO2 with a suitable detergent is used. Use of H2O2 in place of Cl2 for bleaching clothes gives better results and makes use of lesser amount of water.
3. Soap is 100% biodegradable. Thus, it can be used in place of detergents. Nowadays biodegradable detergents are also available. Thus, these are used in place of non-biodegradable detergents.
4. CNG (Compressed Natural Gas) should be used because it causes least pollution. Along with this, use of electrical automobiles can also reduce the consumption of petrol and diesel.
![]()
Question 13.
Differentiate between London classical smog and photochemical smog.
Answer:
Differentiate between London classical smog and Photochemical smog
| Classical smog | Photochemical smog |
| 1. This type of smog was first observed in London in 1952. | This type of smog was first observed in Los Angeles in 1950. |
| 2. It is formed by the reaction of moist SO2 with H2SO4, which collects as particulate matter. | It is formed by photochemical reaction between NO2 and hydrocarbon. |
| 3. It is formed by smoke and fog. | It does not contain smoke and fog. |
| 4. It is formed in the morning in winter. | It is formed in warm sunny climate at day time. |
| 5. It causes lung diseases. | It causes irritation in eyes. |
| 6. It shows reducing character. | It shows oxidizing character. |
Question 14.
(a) What are the main sources of soil pollution?
(b) How can Environmental pollution be controlled?
Answer:
(a) Main sources of soil pollution are :
- Industrial waste.
- Insecticides, pesticides and herbicides.
- Fertilizer, DDT, BHC, NaClO3, Na3ASO3 etc.
- Radioactive products.
(b) Environmental pollution includes domestic waste, industrial waste etc. which can be controlled by the following methods :
- By the recycling of glass, plastic, iron, polythene, paper etc.
- By burning.
- By management of waste.
- By the use of Green chemistry.
- By sewage treatment.
- By awareness.
- By planting more trees.
- By adopting control tools and techniques.
- By installing high chimneys fitted with equipments to discharge pollutants.
Mechanical Properties of Fluids Class 11 Important Questions Objective Type
1. Choose the correct answer:
Question 1.
Source of non-pollution energy :
(a) Fossil fuel
(b) Sun
(c) Gasoline
(d) Nuclear energy.
Answer:
(b) Sun
Question 2.
Which radiations manufacture O3 :
(a) Ultraviolet
(b) Visible
(c) Infrared
(d) Radio waves.
Answer:
(a) Ultraviolet
Question 3.
Which radiations provide greenhouse effect:
(a) Infrared
(b) Visible
(c) Ultraviolet
(d) X-rays.
Answer:
(a) Infrared
Question 4.
PAN is responsible for :
(a) Depletion of ozone layer
(b) Smog
(c) Acid rain
(d) Poisonous food.
Answer:
(b) Smog
![]()
Question 5.
Which is not an air pollutant:
(a)H2
(b) H2S
(c) NOx
(d) O3.
Answer:
(a) H2
Question 6.
Air pollutant released from Jet aeroplanes in the form of an air pollutant:
(a) Photochemical oxidant
(b) Photochemical reductant
(c) Aerosol
(d) Physical pollutant.
Answer:
(c) Aerosol
Question 7.
Is not present in acid rain :
(a) H2SO4
(b) HNO3
(c) H2SO3
(d) CH3COOH.
Answer:
(d) CH3COOH.
Question 8.
Is responsible for disease of lungs :
(a) O2
(b) N2
(c) CO2
(d) SO2.
Answer:
(d) SO2.
Question 9.
03 is manufactured in :
(a) Troposphere
(b) Stratosphere
(c) Mesosphere
(d) Thermosphere.
Answer:
(b) Stratosphere
Question 10.
For acid rain ‘sink’ is :
(a) Leaves
(b) Reservoir
(c) Limestone
(d) CO2.
Answer:
(c) Limestone
![]()
Question 11.
Primary pollutant is :
(a) SO2
(b) NO2
(c) N2O
(d) NO.
Answer:
(d) NO.
Question 12.
Most dangerous is :
(a) Smoke
(b) Dust
(c) Smog
(d) NO.
Answer:
(c) Smog
Question 13.
Which of the following easily combine with haemoglobin:
(a) CO
(b) NO
(c) O2,
(d) CO2.
Answer:
(b) NO
Question 14.
Aromatic compound which is found in the form of particles :
(a) Benzene
(b) Toluene
(c) Nitrobenzene
(d) Polyring hydrocarbon.
Answer:
(d) Polyring hydrocarbon.
Question 15.
Following disease is caused due to depletion of ozone :
(a) Blood cancer
(b) Lung cancer
(c) Skin cancer
(d) Chest cancer.
Answer:
(c) Skin cancer
Question 16.
Photochemical smog is formed :
(a) In summer mornings
(b) In winter days
(c) In rainy days in the morning
(d) In day time in rains.
Answer:
(b) In winter days
Question 17.
Which of the following compound does not form photochemical smog:
(a) NO
(b) O3
(c) CxHy
(d) SO2.
Answer:
(d) SO2.
Question 18.
Main pollutant hydrocarbon is :
(a) Methane
(b) Ethane
(c) Propane
(d) Butane.
Answer:
(a) Methane
![]()
Question 19.
Which of the following acid is in abundance in acid rain :
(a) HNO3
(b) H2SO4
(c) HCl
(d) H2CO3.
Answer:
(b) H2SO4
Question 20.
Which of the following is not a pollutant:
(a) NO2
(b) CO2
(c) O3
(d) (CxHy).
Answer:
(b) C02
Question 21.
Which of the following is not a greenhouse gas :
(a) CO2
(b) CH4
(e) CFCs
(d) O2.
Answer:
(d) O2.
Question 22.
Region near earth’s surface is :
(a) Thermosphere
(b) Mesosphere
(c) Stratosphere
(d) Troposphere.
Answer:
(c) Stratosphere
Question 23.
Main source of CO pollutant is :
(a) Industrial process
(b) Means of transport
(c) Earthquake
(d) Volcano.
Answer:
(b) Means of transport
Question 24.
Marble is the main sink of:
(a) Metallic pollutant
(b) NH3 pollutant
(c) Acid pollutant
(d) None.
Answer:
(c) Acid pollutant
![]()
Question 25.
Which of the following is not a Nobel Prize winner in 2005 :
(a) Y. Chadwin
(b) R.H. Grooms
(c) R.R. Shark
(d) Polythene.
Answer:
(d) Polythene.
Question 26.
Is correct for photochemical smog :
(a) It is reducing in nature
(b) Is formed in winter season
(c) Is a mixture of smog and fog
(d) Cause irritation in eyes.
Answer:
(d) Cause irritation in eyes.
2. Fill in the blanks:
1. The air pollutant released by jet aeroplanes in the form of fluro carbon is …………… .
Answer:
aerosol
2. D.D.T. is …………… poisonous pollutant as compared to B.H.C.
Answer:
more
3. 03 is formed in the …………… level of atmosphere.
Answer:
stratosphere
4. A definite tolerable level of pollutants in the environment is expressed by …………… .
Answer:
TLV
5. Maximum air pollutants are present in …………… level of the atmosphere.
Answer:
troposphere
6. Ozone layer prevent us from …………… rays.
Answer:
ultraviolet
7. Oxides of …………… and …………… cause acid rain.
Answer:
nitrogen, sulphur
8. …………… is the main cause of ozone layer depletion.
Answer:
C.F.C
9. …………… gas is responsible for green house effect.
Answer:
CO2
10. Components which produce pollution are called …………… .
Answer:
pollutant
11. …………… causes harmful effect on lungs.
Answer:
Photochemical smog
12. SO2 pollutant is responsible for the disease of …………… .
Answer:
Lungs
13. …………… is responsible for ultraviolet light.
Answer:
Skin cancer
14. …………… also cause harm to ozone layer.
Answer:
Methane.
3. Match the following:
I.
| ‘A’ | ‘B’ |
| 1. Ozone Hole | (a) Fly ash |
| 2. Particulate | (b) CFC |
| 3. Greenhouse effect | (c) Smog |
| 4. PAN | (d) Infrared radiations |
| 5. Fluorosis | (e) Bone disease. |
Answer:
1. (b) CFC
2. (a) Fly ash
3. (d) Infrared radiations
4. (c) Smog
5. (e) Bone disease.
![]()
II.
| ‘A’ | ‘B’ |
| 1. Smog | (a) Water pollution |
| 2. Main component of acid rain | (b) Depletion of ozone layer |
| 3. COD or BOD | (c) Aerosol |
| 4. Chlorofluorocarbon | (d) SO2 |
Answer:
1. (c) Aerosol
2. (d) SO2
3. (a) Water pollution
4. (b) Depletion of ozone layer.
4. Answer in one word/sentence :
1. Write the definition of pollutants.
Answer:
Chemicals which affect man, animal and plant kingdom are called pollutants.
2. Write the names of two air pollutants.
Answer:
SO2, SO3.
3. Name two pollutants which depletes ozone layer.
Answer:
- Cycle of nitric oxide (NO) and
- C.F.C. (Chlorofluorocarbon) in which CFC is main.
4. In which region ozone is found?
Answer:
Ozone is found in stratosphere region.
5. Tell two health problems caused by SO2.
Answer:
- SO2 affects the respiratory canal and lungs due to which various health diseases are caused like cancer also.
- Acid rain caused due to SO2 produces boils on the skin.
6. What is Acid rain?
Answer:
Various gaseous pollutant present in the atmosphere like SO2, SO3, NO2, NO dissolve in raindrop.
These drops fall with rain and are called acid rain.
SO2 + H2O → H2SO3
SO3+H2O → H2SO4.
7. Name any two greenhouse gases.
Answer:
CFC and CO2.
![]()
8. What is PAN?
Answer:
Peroxy acyl nitrate which is photochemical smog.
9. What is CFC?
Answer:
It is chlorofluorocarbon which is the main cause of depletion of ozone layer.
10. What is green chemistry?
Answer:
A technique to check pollution in which such chemical reactions are suggested which do not cause pollution and if pollution spreads then it can be destroyed. This is called green chemistry.
11. What is TLV?
Answer:
A definite value of pollutants can be tolerated. This is expressed by TLV. TLV means ‘Threshold Limit Value’.
12. What are particulate pollutants?
Answer:
The pollutants mixed up with air in liquid or solid state in such a manner that the remain suspended for a long time are called particulates.
13. What is specimasion?
Answer:
Many pollutants can be made from an element. The method of determining which of the product is more dangerous is called specimasion.
14. What is sink?
Answer:
Sink is that in which the substance totally gets consumed and then also there is no effect on sink. ‘
15. Name the biggest sink of the earth.
Answer:
The biggest sink of the earth is the sea.
16. What is greenhouse effect?
Answer:
Greenhouse effect: The heating of atmosphere due to absorption of infrared radiation by carbon dioxide and other gases is called greenhouse effect.
17. Explain the mechanism of acid rain.
Answer:
Due to combustion of fuels various pollutant particles of all gas pollutants in the form of smoke spread. Due to very high temperature of industries and other fuels N2 and 02 combine and spread in the atmosphere as oxides of nitrogen. These various gases fall on the earth as rain drops which is called acid rain.
18. Gas leaked in 1984 in Bhopal gas tragedy is.
Answer:
CH3-N = C= O Methyl isocyanate
19. Name the main component responsible for depletion of ozone layer.
Answer:
Chlorofluorocarbon.
20. Name the person who started “Chipko Andolan” for forest conservation.
Answer:
Sunderlal Bahuguna.
21. Write name of disease caused by water pollution.
Answer:
Water pollution causes gastrointestinal diseases such as cholera, typhoid, dysentry, gastroenteritis, polio, hepatitis etc.
22. Name the important medicinal plant used to remove environmental pollution and also useful in skin cancer.
Answer:
Neem (Azaderecta indica).
23. Which gas is the cause of greenhouse effect?
Answer:
Carbon dioxide (CO2).
24. Which is the main sink of CO pollutant?
Answer:
Organisms present on earth.
25. Write the name of four methods used in Green chemistry.
Answer:
- Use of sunlight,
- Micro-oven,
- Micro-waves,
- Use of Sound waves and Enzyme.