Students get through the MP Board Class 11th Chemistry Important Questions Chapter 11 The p-Block Elements which are most likely to be asked in the exam.
MP Board Class 11th Chapter 11 The p-Block Elements
The p-Block Elements Class 11 Important Questions Very Short Answer Type
Question 1.
How would you explain the higher stability of BC13 in comparison to TiCl3?
Answer:
Boron exhibits only +3 oxidation state, thus it forms a stable compound BCl3. Down the group, inert pair effect gradually becomes more effective due to which+1 oxidation state of Thallium is more stable than its +3 oxidation state. That is why BCl3 is more stable thanTiCl3.
Question 2.
Why does Boron trifluoride act as a Lewis acid?
Answer:
Due to electron deficiency, BF3 acts as a strong Lewis acid. It easily reacts with Lewis base and completes its octet towards boron.
![]() .
.
Question 3.
Giving the examples of BCl3 and CCl4 compounds, explain their behaviour towards water.
Answer:
BCl3 is an electron deficient molecule. It easily accepts an electron pair from water and forms boric acid (H3BO3) and HCl.
BCl3 + 3H2O → H3BO3 + 3HCl
CCl4 is an electron sufficient molecule in which 4 does not undergo hydrolysis.
Question 4.
What is the reason that caustic base like NaOH is not stored in aluminium containers?
Answer:
On keeping caustic base like NaOH in aluminium containers, aluminium dis-solves in base to form sodium meta aluminate, therefore base is not stored in aluminium containers.
2Al + 2NaOH + 2H2O → 2NaAlO2 + 3H2.
Question 5.
At normal temperature, aluminium does not react with water. Why?
Answer:
In presence of air, a transparent protective oxide layer is formed on the surface of aluminium. Due to this layer, at normal temperature, aluminium does not react with water.
![]()
Question 6.
Among iron and aluminium, aluminium is more reactive as compared to iron but iron easily gets rusted than aluminium. Why?
Answer:
In presence of air, a transparent non-porous protective layer of oxide is developed on aluminium surface due to which at normal temperature it does not react with oxygen and moisture present in air. Whereas a porous oxide layer is formed on iron surface, due to which reactivity of iron increases. Therefore iron easily gets rusted.
Question 7.
Write the formula of double salt or Alum.
Answer:
General formula of double salt is R2SO4.M2(SO4)3 where R is a monovalent metal like Na, K, Rb, Cs or NH4 radical and M is a trivalent metal like Fe+3, Al+3 or Cr+3.
Example: K2SO4.Al2(SO4)3.24H2O (Potash Alum).
Question 8.
Name the ores of aluminium. Give their formulae.
Answer:
Ores of aluminium are as follows :
1. Bauxite: Al2O3.2H2O
2. Diaspore: Al2O3.H2o
3. Cryolite: Na3AlF6
4. Alunite: K2SO4.Al2(SO4)3.2Al(OH)3
5. Corundum: Al2O3
6. Felspar: K2O.Al2O3.SiO3.
Question 9.
What happens when (Give chemical equation):
(a) Aluminium chloride is heated, (b) Alum is heated.
Answer:
(a) On heating aluminium chloride :
2AlCl3.6H2O → Al2O3 +6HCl+ 3H2O
Thus on heating anhydrous aluminium chloride cannot be obtained.
(b) On heating alum to 200°C a porous substance is obtained.
K2SO4. Al2 (SO4)3.24H2O → K2O + Al2O3 + 4 SO2 + 24H2O
Question 10.
Aluminium is a strong reducing agent. Why?
Answer:
Elements which donate electrons and form cations are known as reducing agents. Reducing nature of an element depends on its standard electrode potential. Higher the negative value of standard electrode potential of an element higher is its reducing character. Standard electrode potential of AI is -1.67, therefore aluminium acts as a strong reducing agent.
Question 11.
Why is Gallium a liquid at room temperature?
Answer:
In solid state crystalline structure of gallium is such that its lattice energy is very less and at low-temperature metallic bond between its atoms start breaking. Therefore gallium is obtained in liquid state at low temperature.
Question 12.
Boron does not form trivalent ion. Why?
Answer:
Due to small size of boron, its ionization energy is high and value of third ionization is more than that of first and second ionization energy. Therefore, it is not possible to remove or donate three electrons. Therefore it does not form trivalent ion.
![]()
Question 13.
Is boric acid a protic acid ? Explain.
Answer:
Boric acid, is not a protic acid, because it does not give proton on ionization in water. It accepts electron from the hydroxyl ion of water and behaves as a Lewis acid and releases H+ ion at the end.
B(OH)3 + HOH → [B(OH)4] + H+.
Question 14.
Give the reactions representing amphoteric behaviour of aluminium.
Answer:
Al dissolves both in acid and base and liberate dihydrogen. This behaviour of it, is amphoteric.
2Al(s) + 3H2SO4(aq) → Al2 (SO4)3(aq) + 3H2(g)
2Al(s) + 2NaOH(aq) + 6H2O(l) → 2Na+ [Al(OH)4]–(aq) + 3H2(g)
Sodium tetrahydroxoaluminate (III).
Question 15.
Write resonance structures of CO3-2 and HCO3–.
Answer:
Resonance structures of CO3-2
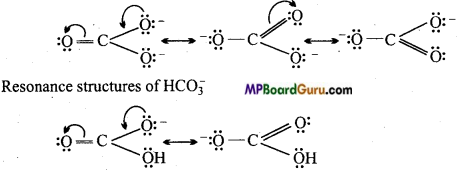
Question 16.
Why is melting and boiling point of boron extremely high?
Answer:
Crystals of boron are formed by the development of covalent bonds between its atoms. Two atoms combine to form icosahedron network which contain 20 triangular faces and 12 comers. It makes boron extremely hard, therefore melting and boiling point of boron is extremely high.
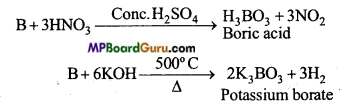
Question 17.
Normally, boron does not react with acid or base under what conditions does it react with acid or base ?
Answer:
Normally, boron does not react with acid or base but if the acid is strongly reducing then boron reacts with it at high temperature and forms boric acid. Similarly, it reacts with base at high temperature to form borate.
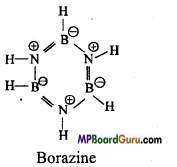
Question 18.
What is inorganic benzene? Le
Answer:
Borazine is known as inorganic benzene. Its chemical formula is B3N3H6. Structure of borazine is like benzene. It has cyclic and planar hexagonal structure.
Question 19.
What is corundum? Borazine
Answer:
Al is found in more than one crystalline forms. Its hardest crystalline form is known as corundum which acts as an abrasive.
Question 20.
State the names of ores ot boron. Give their formulae.
Answer:
Ores of boron are as follows:
- Borax – Na2B4O7.1oH2O
- Kernite – Na2B4O7.2H2O
- Colemanite – Ca2B6O11 .2H2O
- Orthoboric acid – H3BO3
Question 21.
Justify that Tl+3 is an oxidizing agent whereas Al+3 is not?
Answer:
Due to inert pair effect, in boron family, stability of + I oxidation state increases on moving down the group whereas stability of +3 oxidation state decreases. Therefore, Tl+1 is more stable than Tl+3 Thus,
Tl+3 +2e – → Tl+1
By the reaction, it is clear that Tl+3 is getting reduced to Tl+1, therefore Tl+3 is an oxidizing agent but in Al, Al+3 oxidation state is possible and not Al+1, therefore it is not possible for Al+3 to be an oxidizing agent.
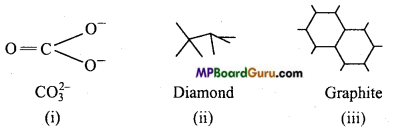
Question 22.
What is the hybridized state of carbon in
(i) CO32-
(ii) Diamond,
(iii) Graphite?
Answer:
Hybridization of carbon in CO32-, diamond and graphite are sp2, sp3 and sp2 respectively.
Question 23.
B-Cl bond has a dipole moment, but dipole moment of BCl3 molecule is zero. Why?
Answer:
In BCl3, boron is sp2 hybridized, due to which geometry of BCl3 molecule is trigonal planar. It has a symmetrical shape and resultant dipole moment of symmetrical molecules is zero (because all dipole moments become negligible due to symmetry of molecules).
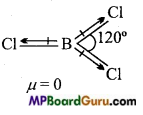
Thus, dipole moment of BCl3 is zero.
![]()
Question 24.
What happens when boron is treated with caustic soda?
Answer:
At ordinary temperature, boron does not show any reaction with base but it reacts with caustic soda NaOH or caustic potash (KOH) to form borate and liberate H2 gas.

Question 25.
Explain disproportionation reaction with example.
Answer:
Gallium exhibits +1 and +3 oxidation states. +3 oxidation state of gallium is more stable, therefore compound of gallium of+1 oxidation state oxidizes to compound of +3 oxidation state.
3GaCl → 2Ga + GaCl3.
Question 26.
Boric acid acts as a Lewis acid, not as a protic acid. Why?
Answer:
In boric acid, octet of central metal boron is not complete. Its valence shell has 6 electrons, so it needs a lone electron pair to complete its octet. Due to being an electron pair acceptor, boric acid acts as a Lewis acid. It liberates H+ ion on reacting with water.
H3BO3 or B(OH)3 + H2O ⇌ [B(OH)4]–+ H+
Question 27.
How is borax obtained from colemanite?
Answer:
On boiling colemanite with concentrated sodium carbonate solution, borax is obtained.
Ca2B6O11 + 2Na2CO3 → Na2B4O7 + 2NaBO2 + 2CaCO3
On concentrating the solution, crystals Of borax are obtained on passing carbon dioxide in the mother liquor borax is obtained.
4NaBO2 + CO2 → Na2B4O7 + Na2CO3
Question 28.
What is the effect of heat on boric acid?
Answer:
On heating boric acid to 100°C temperature, metaboric acid is obtained which on heating to high temperature forms boric anhydride.
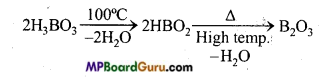
Question 29.
Cryolite is used in the extraction of aluminium by alumina. Why?
Answer:
Melting point of pure alumina is very high 2050°C, but in presence of cryolite and fluorspar, it melts at 870°C. This way, cryolite reduces the melting point of alumina and also acts as an electrolyte.
Question 30.
How will you justify that diamond and graphite are the allotropes of carbon?
Answer:
On combustion of diamond and graphite in the presence of air, C02 gas is re-leased which on passing through lime water turns it milky. This proves that diamond and graphite are the allotropes of carbon.
Cdiamond + O2 →CO2
Cgraphite + O2 → CO2
Question 31.
What will happen if a piece of diamond is dropped on burning charcoal?
Answer:
If a piece of diamond is dropped on burning charcoal, then it will bum completely and only CO2 gas is obtained and after combustion no residue is left which proves that diamond is the purest form of carbon.
Question 32.
Write the uses of carbon monoxide.
Answer:
- It is the main constituent of water gas (CO + H2) and producer gas (CO + N2).
- It is used for the preparation of some metal carbonyls.
- Carbon monoxide is used as reducing agent.
Question 33.
Diamond is less abundant than graphite in nature. Why?
Answer:
Diamond is formed due to conversion of carbon in molten state at extremely high pressure into crystalline form. But this stage in nature is very rare, therefore diamond is less abundant than graphite.
Question 34.
What is dry ice? Write its use.
Answer:
Solid carbon dioxide is known as dry ice because its crystals appear as ice and they do not wet paper or clothe. It gets converted to solid state at -78.5°C without forming a liquid. It is used as a coolant to prevent food materials from decay and as an anaesthetic in surgery.
![]()
Question 35.
What is carborundum? Write its main use.
Answer:
Structure of silicon carbide is hard like diamond, it is known as carborundum. It is used for sharpening and grinding the metals.
Question 36.
Write name and structural formulae of organic compounds used as refrigerant, anaesthetic and solvent.
Answer:
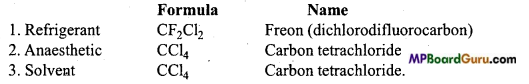
Question 37.
Write the uses of graphite.
Answer:
Uses of graphite :
- It is a good conductor of electricity. So it is used as an electrode in dry cells, electric arcs etc.
- Pencil, black paint, black ink is prepared from it.
- Due to its lubricating property, it is used at high temperature to maintain the machines smooth.
Question 38.
Write the names of various varieties of coal.
Answer:
On the basis of carbon content, coal is of the following varieties :
- Peat: Contains 60% carbon,
- Lignite: Contains 70% carbon,
- Bituminous : Contains 70% carbon,
- Anthracite : Contains 90% carbon.
Question 39.
Write the uses of diamond.
Answer:
Uses of diamond :
- As valuable jewellery,
- Used in cutting glass,
- In drilling rocks,
- In polishing gems.
Question 40.
Carbon dioxide is acidic in nature. Explain with equation.
Answer:
Aquaous solution of carbon dioxide is acidic.
CO2 + H2O → H2CO3 (Carbonic acid)
It turns blue litmus to red and reacts with base to form salt.
2NaOH + CO2 → Na2CO3 + H2O
Ca(OH)2 + CO2 → CaCO3 + H2O.
Question 41.
Why should we not sleep in a closed room with burning chulha (Angithi)?
Answer:
We should not bum chulha (angithi) in a closed room, because the gas coming out of it contains large amount of CO. It enters the body by the process of respiration, combines with haemoglobin and forms carboxyhaemoglobin which creates obstruction in the transportation of oxygen and blood in the body. Therefore, the person can become unconcious and can die also.
![]()
Question 42.
What are carbides?
Answer:
Binery compounds of carbon which it forms with less electronegative or highly electropositive elements than itself are called carbides. These are of various types, like :
- Ionic carbide,
- Metallic carbide,
- Intermediate carbide,
- Covalent carbide.
Question 43.
Write the use of silica gel.
Answer:
Silica gel is a porous, amorphous solid which contains 4% of moisture. It is used as a catalyst in petroleum industry, also in chromatography.
Question 44.
What is thixotropy?
Answer:
On shaking or agitating a liquids, its viscosity decreases temporarily. This property is known as thixotropy.
When SiCl4 is hydrolysed at a high temperature, then the silica obtained this property.
![]()
Question 45.
What are the interstitial carbides?
Answer:
When carbon atoms occupy the interstitial sites of crystal lattice of transition metals, then such carbides formed are called interstitial carbides. They are extremely hard and the melting points are high.
Example: Tungsten carbide, iron carbide.
Question 46.
What are methanides and acetylides?
Answer:
1. Carbides which give methane on hydrolysis are called methanides.
Al4C3 +12H2O →4Al(OH)3 +3CH4
2. Carbides which give acetylene on hydrolysis are called acetylides.
CaC2 + 2H2O → Ca(OH)2 + C2H2.
Question 47.
What are silanes and germanes ?
Answer:
Hydrides of Si and Ge are known as silanes and germanes which are represented by MnH2n+2
where M = Si, Ge. In silanes, value of n is from 1 to 8 whereas in germanes value of n is 1 to 5.
![]()
Question 48.
What is activated charcoal?
Answer:
Charcoal is soft and porous. It absorbs coloured substances and smelling gases. If it is heated in steam to 1100°C, then its absorbing power increases and it is known as activated charcoal.
The p-Block Elements Class 11 Important Questions Short Answer Type
Question 1.
What happens when boric acid is heated?
Answer:
When boric acid is heated, it liberates three molecules of water at various temperatures and forms boron trioxide at the end.
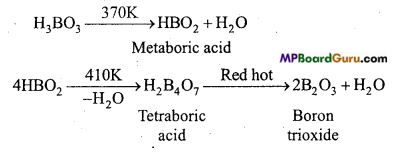
Question 2.
Describe the shapes of BF3 and BH4–. Assign the hybridization of boron in these species.
Answer:
In BF3, three bond pairs are present in boron. Thus, it is sp2 hybridized and of trigonal planar structure, whereas bond pair number in [BH4]– is 4 due to which it is sp3 hybridized and has tetrahedral structure.
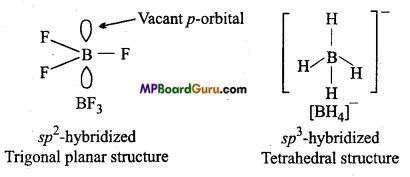
Question 3.
What is alum? State its general formula and use.
Answer:
Initially, double salts of potassium sulphate and aluminium sulphate K2SO4.Al2(SO4)3.24H2O are known as alums. But presently, all double salts with general formula R2SO4.M2(SO4)3.24H2O called alums. Where R = monovalent metal like : Na, K, Rb, Cs, etc. M = Trivalent metal like:PAl, Cr, Fe etc.
Uses :
- In the purification of water,
- In dyeing leather,
- In paper industry,
- In fire extinguishers,
- In dyeing clothes,
- In clotting of blood.
Question 4.
Write the name, composition and uses of four alloys of aluminium.
Answer:
Alloys of aluminium :
- Aluminium bronze: Cu (90%) + Al (10%)
Uses: For making utensils, cheap ornaments, coins. - Magnelium : Mg (10%) + Al (90%)
Uses : Used for making aeroplanes, tools. - duralumin : Al (95%) + Cu (4%) + Mn (0.5%) + Mg (0.5%)
Uses : Used for making airships. - Nickeloy : Al (95%) + Cu (4%) + Ni (1%)
Uses : For making utensils, coins etc.
Question 5.
Aluminium is a weaker conductor of electricity than copper still it is used in electric cables. Why?
Answer:
Copper is a better conductor of electricity than aluminium but aluminium is a light metal and its density is less than copper. This way according to weight aluminium is a better conductor as compared to copper. Therefore, aluminium is used in making electric wire and cable instead of copper.
Question 6.
Boron forms only covalent compounds. Why?
Answer:
Due to small size and high ionization energy of boron, its tendency to form cation is very less. Therefore boron cannot lose three electrons to form trivalent ion. To achieve a stable electronic configuration it shares its electrons with atoms of other elements to form stable compounds. Therefore, boron forms only covalent compounds.
![]()
Question 7.
Halides of boron acts as strong Lewis acids. Why?
Answer:
There are three electrons in the valence shell of boron. When it shares its electrons with three halogen atoms, then also it contains total 6 electrons in its valence shell, it still needs lone electron pair to complete its octet. Therefore, it is an electron-deficient compound and acts as an electron acceptor and accepts electrons from an electron-pair donor compound to form a co-ordinate bond and form an addition compound. Therefore it acts as a strong Lewis acid.
Question 8.
Aluminium cannot be obtained from its ore by the process of reduction. Why?
Answer:
Due to strong electropositivity, aluminium acts as a reducing agent, therefore aluminium can be easily oxidized, on the basis of ionization energy and electron affinity, it is clear that aluminium acts as electron donor, not as an electron acceptor. Therefore, it cannot be reduced. That is why aluminium cannot be obtained from its ore by reduction.
Question 9.
Atomic radius of gallium is less than aluminium. Why?
Answer:
In gallium, there are 10 electrons in d-orbital. Shape of d-orbital is such that its screening effect is less effective due to which attractive force of nucleus for outermost electrons is more. As a result, outermost electrons are strongly attracted by the nucleus due to which atomic radius of gallium decreases. Therefore atomic radius of gallium is less than aluminium.
Question 10.
Suggest a reason as to why CO is poisonous?
Answer:
Carbon monoxide reacts with haemoglobin to form a stable compound named carboxyhaemoglobin. It is 300 times more stable than oxyhaemoglobin. Due to formation of carboxyhaemoglobin the tendency of flow of oxygen in blood decreases which in turn leads to suffocation. Less amount of it may cause headache and more amount of it may lead to death.
Question 11.
State the reason of formation of covalent compounds of boron halides with ammonia and amine.
Answer:
There are three electrons in valence shell of boron. When it shares electrons with three halogen atoms to form boron trihalide then also the valence shell of boron contains total 6 electrons. They still require an electron pair to complete their octet. Therefore, they are electron deficient compounds and act as electron acceptor and accept a lone pair of electron from any electron-pair donor compound like ammonia or amine to form a co-ordinate bond and result in the formation of an addition compound.
Question 12.
Boron family generally exhibit +1 and +3 oxidation state. Why?
Answer:
B5 → 1s2,2s22p1
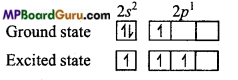
General electronic configuration of all the members of boron family is ns2 np1. In normal state, there is one unpaired electron in p-orbital of its valence shell. But in excited state, one electron of 2s gets excited and goes to 2p-subshell. Thus, in excited state there are three unpaired electrons. Therefore, all the members of boron family exhibit +1 and +3 oxidation state.
Question 13.
Explain inert pair effect in boron family.
Answer:
Members of boron family exhibit +1 oxidation state in normal state and +3 in excited state. On moving down the group, stability of +1 oxidation state increases but that of +3 oxidation number decreases because the two electrons of 5-orbital of valence shell do not participate in bond formation. This is known as inert pair effect.
With the increase in atomic number, electrons enter into d-subshell and shape of d and d-subshells is such that their screening effect is minimum due to which attractive force of nucleus on valence electron increases. This increase in attractive force is more in 5-orbital electrons than in electrons of p-subshell. Therefore 5-orbital electrons do not participate in bond formation.
![]()
Question 14.
How is boric acid obtained from colemanite?
Answer:
Colemanite is dissolved in boiling water and on passing sulphur dioxide gas through it, boric acid and calcium bisulphite are formed. Calcium bisulphite is soluble whereas boric acid crystallizes.
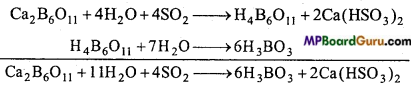
Question 15.
What is borax glass?
Answer:
Anhydrous sodium tetraborate (Na2B4O7) is known as borax glass. It is obtained on heating ordinary borax above its melting point. It is a colourless glass-like substance which absorbs moisture from the environment and gets converted to decahydrate form. It is soluble in hot water, its aqueous solution is alkaline due to hydrolysis. On heating, it swells up to a white opaque substance. Anhydrous substance gives borax glass at 740°C.
Na2B4O7+ 2H2O → H2B4O7 + 2NaOH
Question 16.
Explain the effect of heat on borax.
Answer:
On strong heating borax, its water of Crystallization separates and finally it melts and gets converted to transparent bead.
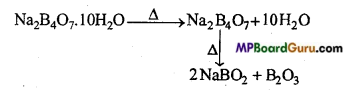
Boric anhydride B2O3 reacts with metallic oxides to form metaborate which has a specific colour. This reaction is known as borax bead test and is used in the detection of basic radicals.
Question 17.
How is excessive content of CO2 responsible for global warming?
Answer:
CO2 is a greenhouse gas. 75% of the solar energy reaching the earth is absorbed by the earth’s surface and the remaining is reflected back into the atmosphere. But the heat released from the hot surface cannot be sent back to space because the C02 present in the atmosphere absorbs this extra heat due to which average temperature of the atmosphere increases. This is known as global warming.
Question 18.
Explain why is there a phenomenal decrease in ionization enthalpy from carbon to silicon?
Answer:
On moving from carbon to silicon in the periodic table, the atomic size increases, thus distance between the outermost electron and nucleus increases. Thus, these electrons feel very less attraction of the nucleus, due to which their removal is very easy. Since size of Si atom is considerably larger due to which the outermost electrons feel less attraction. Therefore, its ionization enthalpy (to remove 1 electron) is less.
Question 19.
How would you explain the lower atomic radius of Ga as compared to Al?
Answer:
Electronic structures of Al and Ga are as follows :
13Al = 1 s2,2s22p6,3s23p1
31 Ga = 1s2,2s2,2p6,3s2,3p63d10,4s24p1
In this screening effect of d-electrons is very less. Thus, on moving from Al to Ga, screening effect of 10 d-electrons is unable to make the nuclear charge ineffective. Thus, atomic radius of Ga, due to effective nuclear charge is less than that of aluminium.
Question 20.
What is borax bead test? Explain.
Answer:
Borax bead test: On heating borax it loses its water of crystallisation. On further heating it forms a glassy bead which contains sodium metaborate and boric anhydride.
Na2B4O7.10H2O → Na2B4O7+10H2O
Na2B4O7 → 2NaBO2 + B2O3
Example: Copper sulphate forms a bluish green metaborate.
CuSO4 → CuO+SO3
CuO + B2O3 →Cu(BO2 )2
Cupric metaborate bead of bluish green colour In reducing flame the colour of the bead is red.
2Cu(BO2)2 →CuBO2 +B2O3 + CUO
Thus, Borax bead test is used in the detection of basic radicals.
Question 21.
How many types of borax are there? Describe briefly.
Answer:
Borax exists in the following three forms :
1. Prismatic borax: It is decahydrate, Na2B4O7.10H2O. It is the ordinary form and is obtained by the crystallization of solution at ordinary temperature.
2. Octahedral borax: It is borax pentahydrate, Na2B4O7.5H2O. It is formed by the crystallization of the solution above 60°C.
3. Borax glass: It is anhydrous sodium tetraborate, Na2B4O7. It is obtained by heating ordinary borax above its melting point. It is a colourless glass like substance. It absorbs moisture from the air and gets converted to decahydrate form. Its aqueous solution is basic.
Na2B4O7 +2H2O → H2B4O7 +2NaOH.
![]()
Question 22.
How is borate radical detected?
Answer:
In the laboratory, for the detection of acidic borate radical BO3-3, salt is heated with ethanol and concentrated sulphuric acid, due to which vapours of ethyl borate are obtained. These vapours bum with a green core flame. Actually, the salt first gets converted to boric acid, which reacts with ethanol to form triethyl borate.
H3BO3 +3C2H5OH →B(OC2H5)3 + 3H2O.
Question 23.
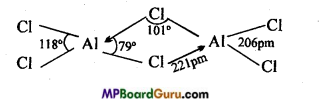
Explain the structure of aluminium chloride.
Answer:
Aluminium chloride is actually obtained in the form of dimer Al2Cl6. There are three electrons in the valence shell of Al, when these three electrons are shared with three chlorine atoms, AlCl3 is formed but there are total 6 electrons in the valence shell of Al. To complete its octet, it requires one lone electron pair. In tfcis conation, Al of AlCl3 electron pair from chlorine of AlCl3 and complete its octet.
Question 24.
What is the reason that AlF3 is insoluble in anhydrous HF but becomes soluble on the addition of NaF Why?
Answer:
Strength of hydrogen bond in HF is more, due to which HF is comparatively a weak acid and cannot release F ion. On adding NaF, due to complete ionization concentration ofF ion considerably increases by which due to formation of complex ion AlF6-3 by AlF3 it dissolves.
HF → H+ + F–
3NaF → 3Na++ 3F–
AlF3 + 3F– → AlF6-3
AlF6-3 + 3Na+ → Na3 [AlF6].
Question 25.
In some reactions thallium resembles aluminium, whereas in others it resembles with group-1 metals. Support this statement by giving some evidences.
Answer:
Both thallium and aluminium are the elements of group-13. General electronic configuration of their valence shell is ns2 np1. Aluminium exhibits only +3 oxidation state. Like aluminium, thallium also exhibits +3 oxidation states in some of its compounds.
Example: Tl2O3, TlC3 etc. Like aluminium, thallium also forms octahedral ion like [AlF6]-3– and [TlF6]3-.
Like alkali metals of group-1, due to inert pair effect, thallium exhibits +1 oxidation state.
Example: TlCl, Tl2O etc. Like alkali metal hydroxides, TlOH is also soluble in water and its aqueous solution is strongly basic. Tl2SO4, like alkali metal sulphates form alum and Tl2CO3, like alkali metal carbonates soluble in water.
Question 26.
Explain the difference in properties of diamond and graphite on the basis of their structure.
Answer:
Differences in properties of Diamond and Graphite
| Diamond | Graphite |
| 1. In this C is sp3 hybridized. | In this C is sp2 hybridized. |
| 2. Its geometry is three-dimensional tetrahedral. | Its geometry is two-dimensional hexagonal layers. |
| 3. It is hardest substance with high density and high boiling point. | It is a soft slippery substance with low density and high boiling point. |
| 4. It is a bad conductor of electricity (due to absence of free electrons). | It is a good conductor of electricity (fourth electron is free). |
| 5. It is used in cutting glass, jewellery and as an abrassive. | It is used as a lubricant, in the manufacture of electrodes, in making pencils, crucibles (due to high melting point) etc. |
Question 27.
Explain back bonding with example.
Answer:
There are total 6 electrons in the valence shell of boron trifluoride. Being an electron acceptor, BF3 acts as a Lewis acid. It should be a strong Lewis acid but it acts as a weak Lewis acid.
BF3 is a symmetrically planar molecule due to sp2 hybridized boron in BF3. In this molecule, one 2pz orbital of boron remains completely vacant. On the other side, there are 2 electrons in 2pz orbital of fluorine. In this situation, due to overlapping between 2pz orbital of boron and 2pz orbital of fluorine a bond can be formed. This is known as back bonding.
Question 28.
1. BCl3 is stable but B2Cl6 does not exist whereas AlCl3 is unstable.
2. AlCl3 is unstable, Al2Cl6 is stable. What is the cause?
Answer:
1. BCl3 is stable because of the presence of electrons in its valence shell but due to back bonding, the various resonating structure provide stability to BCl3 due to resonance. But B does not contain vacant d orbital, therefore, it cannot accept the electron pair donated by chlorine, therefore formation of B2Cl6 is not possible.
2. In the valence shell of AlCl3, there are total 6 electrons. Due to incomplete octet, AlCl3 is unstable but in Al2Cl6 dimer, vacant d orbital of aluminium can accept the lone electron pair donated by chlorine to complete its octet. Therefore Al2Cl6 is stable.
![]()
Question 29.
Write the formula and two uses of the following compounds :
1. Borax,
2. Boric acid.
Answer:
1. Borax:
Formula: Na2B4O7.10H2O
Uses:
- Due to its resisting character, it is used in the preparation of medicinal soaps.
- In the manufacture of spectacle glass (boro glass).
2. Boric acid :
Formula: H3BO3
Uses :
- Boric acid is used in the manufacture of enamel and in brightening utensils.
- Due to its antiseptic nature, boric acid is used to wash eyes.
The p-Block Elements Class 11 Important Questions Short Answer Type – II
Question 1.
Rationalise the given statements and give chemical reactions :
(a) Lead (II) chloride reacts with Cl2 to give PbCl4.
(b) Lead (IV) chloride is highly unstable towards heat.
(c) Lead is known not to form an iodide Pbl4.
Answer:
(a) Due to inert pair effect, Pb is more stable in +2 oxidation state than in +4 oxidation state.
Thus, lead (II) chloride does not give lead (IV) chloride on reacting with chlorine.
(b) Lead (IV) chloride dissociate on heating and gives lead (II) chloride and Cl2 because lead is more stable in +2 oxidation state than in +4 oxidation state.
2PbCl4(l) →PbCl2(s) + Cl2(g)
(c) Due to strong reducing tendency of Pb+4 ions and reducing tendency of F– ion, Pbl4 does not exist.
Question 2.
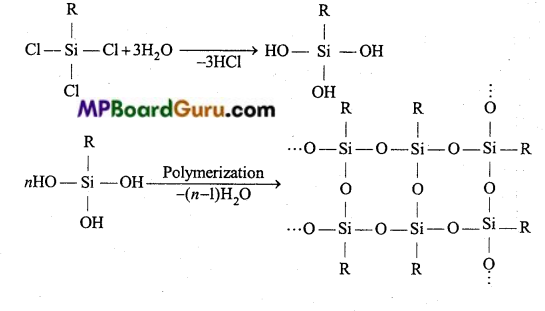
If starting material for the manufacture of silicones is RSiCl3. Write the structure of the product formed.
Answer:
By the hydrolysis of alkyl trichlorosilane followed by condensation polymerization, chain polymer (silicone) is obtained.
Question 3.
What is catenation? In which element this property is maximum and why?
Answer:
The property of linking with atoms of its own kind is called catenation. This property is maximum in carbon because carbon atom is small in size and its electronegativity is high. Bond energy of C-C bond (355.3 kJ mol-1) is maximum. Thus, it possess the tendency to form long carbon chains.
Question 4.
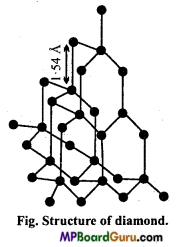
Give structure of Diamond.
Answer:
Diamond is a tetrahedral molecule, forming three- dimensional network. Each carbon has sp3 hybridization and is linked with four other carbon atoms by strong covalent bonds as shown in the Fig. It is clear from the figure that each carbon is situated at the centre of a regular tetrahedron and other four carbon atoms are present at the four corners of it. Experimental data proves this structure. In this structure, C-C bond length is 154 A (154 picometer).
Hence, in structure of diamond there is three-dimensional network of strong covalent bonds. It is for this reason that the diamond is the hardest substance known and it has a very high melting point (m.p. of diamond is 3843 K). All the valencies of carbon take part in the formation of C – C bonds and therefore, diamond is a bad conductor of electricity.
Question 5.
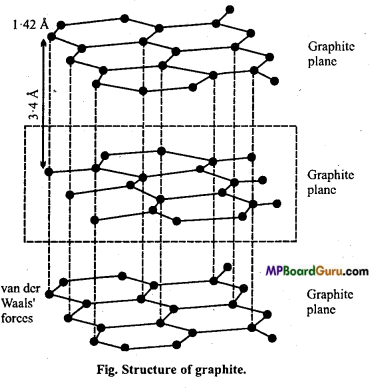
Give structure of Graphite.
Answer:
Structure: In graphite, each carbon is sp2 hybridized. Each carbon is linked with three other carbon atoms by single covalent bonds. The fourth electron of each carbon remains free and this accounts for good conductivity of graphite. The C – C bond length in graphite is 1.42Å (142 picometer). All the three sp2 hybridized orbitals lie in the same plane (are planar). Hence, in graphite carbon atoms form a hexagonal ring and lie in the same plane.
These rings combine together to form a plane. In graphite, there are many such planes which lie one over the other at a distance of 3.4Å. Weak van der Waals’ forces are acting in between these planes. Due to this reason, it is possible to slip one plane of graphite over other and this is responsible for lubricating property of graphite and its soft nature.
Question 6.
Explain industrial method of preparation of Artificial graphite with chemical equation.
Answer:
Preparation of artificial graphite: Graphite is obtained by Acheson (Edward G. Acheson) method in electric arc furnace. Mixture of coke and sand is heated in electric furnace. The furnace is fitted with carbon electrodes which are connected together with a thin rod of carbon as shown in the fig.
Electric current is passed. The temperature reaches to 3000°C. Iron oxide or silica acts as catalyst in the reaction.
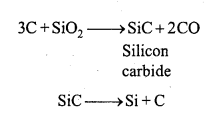
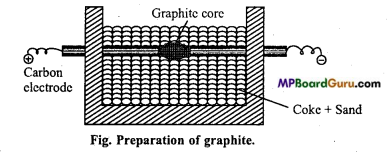
Question 7.
Explain the method of preparation of artificial diamond.
Answer:
A mixture of sugar, charcoal and iron oxide is taken in a crucible and heated in an electric furnace to a temperature of 3000°C. Then, it is placed in molten lead. Temperature of molten lead is less than iron due to which iron starts solidifying. As a result of pressure carbon starts separating in the form of small crystals of diamond. Iron is separated by dissolving in HCl. This way, artificial diamond is obtained.
Question 8.
Explain Labell and van’t Hoff law related to valency of carbon atom. Or, What do you understand by symmetrical tetrahedral structure of carbon?
Answer:
Atomic number of carbon is 6. On this basis, 2 electrons are in first shell and 4 electrons in the second shell. Thus, its valency is four. According to Labell and van’t Hoff, if we consider carbon atom to be situated at the centre of tetrahedron and the four arms of tetrahedron representing the four valencies of carbon, then the value of angle between any two valencies is 109°28’. By Henry’s experiments, carbon valencies are symmetrically arranged. It is in tetrahedral form in space. These are not in a plane.
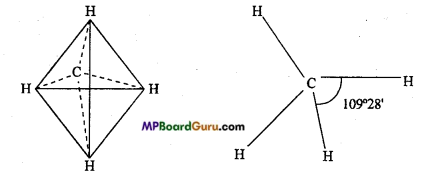
Question 9.
Give the cause of lubricanting property of graphite.
Answer:
In graphite, carbon atoms link with each other by covalent bonds and form a hexagonal network. These rings mutually form a layer. In graphite, many such layers lie one over the other at a distance of 3.4 Å and van der Waals’ force are bonded to each other by weak due to which they can easily slide over each other. As a result, graphite is soft and its melting point is high. Therefore graphite is used as a lubricant for such machines which become hot when used.
![]()
Question 10.
Diamond is used in cutting tools. Why?
Answer:
In diamond, each carbon atom is in sp3 hybridized state and each carbon is linked with other carbon atoms by strong covalent bonds. Thus a three-dimensional tetrahedral structure is formed in diamond which is very strong. Therefore, diamond is hardest known element and therefore it is used in cutting tools.
Question 11.
Diamond has a specific shine (brilliance). Why? Or, Diamond is used in making jewellery, why?
Answer:
High refractive index and total internal reflection makes diamond shine and appear beautiful. Therefore, diamond is extremely shiny and is used in making costly jewellery.
Question 12.
Graphite is soft and diamond is hard. Why?
Answer:
In graphite, each carbon atom is in sp2 hybridized state and each carbon atom in graphite link with nearest three atoms in the same plane and form a hexagonal network. Many such planes are loosely held over one another with weak van der Waals’ force due to which layers of graphite can slide over one another. Due to This property graphite is soft.
In diamond, each carbon atom is in sp3 hybridized state in which each carbon atom is joined to other four carbon atoms by covalent bonds and form a tetrahedral three-dimensional structure, therefore diamond is hard.
Question 13.
Diamond is a bad conductor of electricity, whereas graphite a good conductor. Give reason.
Answer:
Both diamond and graphite are the allotropes of carbon and the outermost shell of their atoms contain four electrons. In diamond carbon is sp3 hybridized state and the four electrons of each carbon atom is linked to nearest four carbon atoms by strong covalent bonds. Thus, no free electron is present in any carbon atom, therefore it is extremely hard and a bad conductor of electricity.
In graphite each carbon is in sp2 hybridized state and only three electrons are joined to nearest three carbon atoms by strong covalent bonds and the fourth electron remains free. Therefore, flow of electrons in graphite is easy. As a result graphite is soft, and a good conductor of electricity.
Question 14.
Carbon monoxide has bad effect on our body. Or, Why is CO gas poisonous?
Answer:
CO combines with haemoglobin of blood and forms a stable compound carboxyhaemoglobin.
Haemoglobin + O2 ⇌ Oxyhaemoglobin
In which the ability to carry the oxygen of blood is destroyed due to which the person may become unconscious or even die due to suffocation.
Question 15.
Differentiate physical properties of Diamond and Graphite in a tabular form.
Answer:
Differences in physical properties of Diamond and Graphite
| Physical properties | Diamond | Graphite |
| 1. Appearance | Transparent | Opaque, Black |
| 2. Hardness | Hard | Soft |
| 3. Thermal conductivity | Very low (bad conductor) | Low |
| 4. Electrical conductivity | Bad conductor | Good conductor |
| 5. Density | 3510 kg/m3 | 2250 kg/m3 |
| 6. Use | In jewellery and in cutting tools | As a lubricant, as electrodes. |
Question 16.
What is supercritical liquid?
Answer:
Any gas can be liquefied by increasing pressure at a temperature lower than its criticaL temperature. The pressure at which a gas can be liquefied is known as its critical pressure. But due to sublime nature of CO2, it cannot be converted to liquid state, therefore above critical pressure it can be converted to supercritical liquid. Critical temperature and critical pressure for CO2 is 31°C and 729 atmospheric pressure.
Question 17.
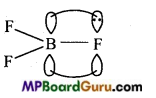
Suggest reasons why the B – F bond lengths in BF3 (130 pm) and BF4 (143 pm) differ.
Answer:
In BF3, boron is sp2 hybridized. It contains vacant 2p-orbital. In BF3, each F contains completely filled 2p-orbital. Since both these orbitals are of similar energy, pπ-pπ back bonding is formed in which one electron pair from completely filled 2p-orbital is transferred to vacant 2p-orbital of boron. This type of bond ] formation is known as back bonding. Thus, some double bond behaviour is found in B – F bond. That is why, bond length of all the three B – F bonds is less.
In [BF4]– ion, boron is sp3 hybridized. It does not contain vacant 2p-orbital due to which back bonding is not found in it. In [BF4]– ion, all the 4 B – F bonds are complete single bonds. Double bonds are smaller than single bonds. Thus, B – F bond length in BF3 (130 pm) is less than in [B4]–(143 pm).
Question 18.
Write the reactions of carbon monoxide which states that it is :
1. Combustible,
2. Unsaturated compound,
3. Reducing agent.
Answer:
1. Combustible: It liberates CO2 gas on combustion in presence of oxygen.
CO+\(\frac{1}{2}\) O2 → CO2
2. Unsaturated compound: Being an unsaturated compound, carbon monoxide forms addition compounds.
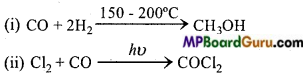
3. Reducing agent: It reduces with metal oxide to metal.
ZnO + CO → Zn + CO2 ↑
Question 19.
Write similarities and dissimilarities in carbon and silicon.
Answer:
Similarities :
- Both carbon and silicon are non-metals.
- Valence shell configuration of both is ns2np2.
- Both show allotropy.
- Both form covalent compounds.
- Covalency of both is four.
- Both possess the property of catenation.
- Oxides of both are acidic.
Dissimilarities:
| Carbon | Silicon |
| 1. Except graphite, carbon is a bad conductor of electricity. | Silicon is a semiconductor. |
| 2. Maximum covalency of carbon is four. | Maximum covalency of silicon is six. |
| 3. Tendency of catenation is more in carbon. | Tendency of catenation is less in silicon. |
| 4. Carbon forms multiple bonds. | Silicon does not form multiple bond. |
| 5. CO is known. | SiO is unknown. |
| 6. CCl4 does not hydrolyse. | SiCl4 undergoes hydrolysis. |
Question 20.
Carbon and silicon show tetravalency whereas Ge, Sn and Pb are bivalent. Why?
Answer:
Due to the small size of carbon and silicon, their ionization energy is very high. Therefore, they do not form ionic compounds by losing electrons but for completing their octet, they share electrons to form covalent compounds and exhibit tetravalency.
Due to large size of Ge, Sn and Pb their ionization energy is very less. Therefore, they can lose electrons to form ionic compounds also and these elements exhibit +2 and +4 oxidation state. Due to inert pair effect on moving from top to bottom in a group, stability of +4 oxidation state decreases, but stability of +2 oxidation state increases. Therefore, they represent +2 oxidation state.
![]()
Question 21.
SnCl4 is a liquid whereas SnCl2 is a solid. Why? Or, Molecular mass of a compound of tin is 189 and of the other is 260, still the first compound is solid whereas the second is a liquid. Why?
Answer:
In SnCl4, Sn exhibits +4 oxidation state and forms covalent compounds, therefore SnCl4 is a liquid whereas in SnCl2, Sn exhibits +2 oxidation state and forms ionic compounds. Therefore, SnCl2 is a solid.
Question 22.
Si, like C does not form graphite-like structure. Why?
Answer:
Si does not form graphite-like structure because :
- Si does not form sp2 hybridized compounds whereas in graphite, carbon is sp2 hybridized.
- Atomic radius of Si is greater than carbon due to which electron affinity, ionization energy etc. is less than carbon due to which Si does not form π -bonds like carbon.
Question 23.
Maximum covalency of carbon is 4, whereas maximum covalency of other members of this group is 6. Why? Or, Carbon does not show higher oxidation state like Si. Why?
Answer:
All the members of carbon family contain 4 unpaired electrons in their valence shell in excited state. They require 4 more electrons to complete their octet. Therefore, they share electrons with other elements and form covalent bond. Therefore, their valency is 4.
In carbon, due to absence of d-orbital higher oxidation state is not possible whereas in other members, higher oxidation state is possible due to presence of vacant d-orbital because due to presence of vacant d-orbital, they can behave as electron acceptor and can accept a lone electron pair from any other electron donor group forming a co-ordinate bond. Therefore, their maximum oxidation state is 6.
Question 24.
CCl4 does not hydrolyse whereas SiCl4 hydrolysis. Why?
Answer:
Due to absence of vacant d-orbital in carbon, its maximum oxidation state is 4 and it cannot increase its oxidation state. Therefore, CCl4 does not hydrolyse.
In Si, due to presence of vacant d-orbital, its maximum oxidation state is 6, therefore SiCl4 can easily accept the lone electron pair donated by water and this way its oxidation state increases, due to which it easily hydrolyse.
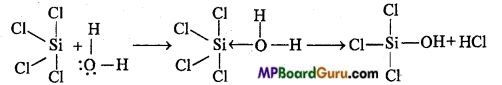
Question 25.
CO2 is a gas, whereas Si02 is a high melting point solid. Why ? Or, Explain the structure of CO2 and SiO2.
Answer:
CO2 has a linear structure. In it carbon is in sp hybridized state and CO2 molecules are attracted by weak van der waals’ force of attraction. Therefore, at normal temperature CO2 is a gas.
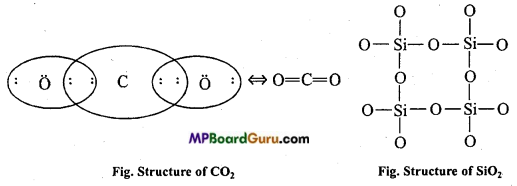
SiO2 is a solid. Its structure is like a three dimentional network. In it, each Si is linked with four oxygen atom in tetrahedral form. Single covalent bond is present between Si and O atom. This single covalent bond is stronger than van der Waals’ force.
Therefore, SiO2 is a hard solid and its melting point is high.
Question 26.
Compare CO2 and SiO2.
Answer:
Comparison of CO2 and SiO2
| Properties | CO2 | SiO2 |
| 1. Physical state | Gas | Solid |
| 2. Structure | Linear monomer | Three-dimensional lattice |
| 3. Melting point | Low | High |
| 4. Reactivity | More reactive | Less reactive |
| 5. Hybridization | sp | sp3 |
| 6. Solubility in water | Soluble | Insoluble |
| 7. Reaction with base | Form carbonate | Form silicate. |
Question 27.
Carbon like other members of its group do not form complex compound. Why?
Answer:
Tendency of an element to form co-ordinate or complex compound depend on the following factors:
1. Small atomic radius,
2. High charge density,
3. Presence of d-orbital.
From the electronic configuration of carbon, it is clear that carbon does not contain vacant d-orbital, therefore it cannot accept electron pair by ligand and form complex compound. Whereas other members of the group contain vacant d-orbital due to which they can easily accept the electron pair donated by ligand and can form coordinate bond. Therefore, they easily form complex compound.
Question 28.
M2+ is strong reducing agent whereas M4+ ion represent covalent character.
Answer:
All the members of carbon family contain 4 electrons in their valence shell and in M4+ state its ionization energy is very high. Therefore, all the elements share their electrons with other elements and form covalent compounds.
Whereas less ionization energy is required to remove two electrons, therefore in +2 oxidation state, they form ionic compounds. Due to the tendency to donate electrons, they behave as reducing agents.
Question 29.
Normally compounds of tin and lead like SnCl2 and PbCl2 are used as reducing agents whereas SnCl, and PbCl4 are used as oxidizing agents. Why?
Answer:
In Sn and Pb, +2 oxidation state is more stable than +4 oxidation state. Thus, Sn4+ and Pb4+ possess the tendency to form Sn2+ and Pb2+ respectively due to which it reduces others. Hence Sn4+ and Pb4+ is oxidising agent and Sn2+ and Pb2+ possess the tendency to form Sn4+ and Pb4+. That’s why it oxidizes others.
Question 30.
Explain the orbital structure of monoxide by carbon.
Answer:
In CO, both carbon and oxygen are in sp-hybridized state.
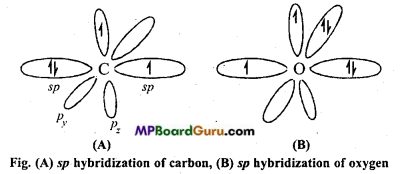
One sp-orbital of carbon overlaps with one sp-orbital of oxygen and form σ-bond. Other orbital of carbon and oxygen contain one-one electron pair which is non-bonding. One electron pz-orbital of carbon undergoes lateral overlapping with one electron of pz – orbital of oxygen to form a π-bond.
Now, there is no electron in p orbital of carbon, whereas there are two electron in py orbital of oxygen. These also undergo lateral overlapping to form a bond which represents co-ordination in Lewis structure.
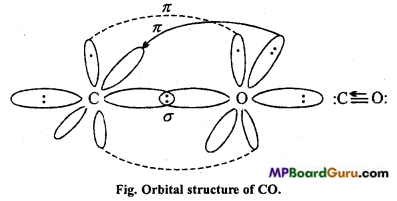
Question 31.
What is Silica garden?
Answer:
In a crystal of saturated aqueous solution of sodium silicate if sand, crystal of copper sulphate, ferrous sulphate, nickel sulphate, cadmium nitrate, manganese sulphate, cobalt nitrate etc. are added then after two three days coloured plants appear to grow in the solution. This is known as Silica garden.
Question 32.
Why does silicon monoxide not form like carbon monoxide ?
Answer:
Carbon can form a π-bond with oxygen after the formation of a σ-covalent bond. Also another vacant 2pz orbital of carbon can undergo overlapping with lone electron pair situated in 2pz orbital of oxygen. Because carbon is so much electronegative that it can accept a lone electron pair by oxygen. Whereas Si is less electronegative and it also has a bigger size due to which it cannot form 3pπ – 2pπ. Therefore, SiO is not possible.
Question 33.
Write balanced equations for the preparation of water gas, carburetted water gas and producer gas.
Answer:
1. Water-gas: This gas is a mixture of carbon monoxide and hydrogen. It is prepared by passing steam over red hot coke.
![]()
2. Carburetted water gas : On passing water gas through hot bricks placed in oil, acetylene and ethylene are formed which mix with water gas to form carburetted water gas. It contains CO = 30%, H2 = 35%, saturated hydrocarbons = 15.20%, hydrocarbon = 10%, N2 = 2.5 – 5%, C02 = 2%.
3. Producer gas: It is a mixture of carbon monoxide and nitrogen. It is obtained by passing limited amount of air on red hot coke.
2C + Air (O2 +N2) →2CO + N2
Question 34.
How is silica gel obtained from silicon tetrachloride?
Answer:
Silicon tetrachloride is obtained by, the reaction of silicon with chlorine.
Si + 2Cl2 →SiCl4
By the hydrolysis of SiCl4, silicon tetrahydroxide is obtained.
SiCl4 + 4HOH → Si(OH)4 +4HCl
This silicon tetrahydroxide is actually silicic acid monohydrate.
Si (OH)4 ⇌ H2SiO3 .H2O
On heating, silicic acid breaks into silica.
H2SiO3.H2O → SiO2 + 2H2O
This silica, is known as silica gel (SiO2 xH2O).
![]()
The p-Block Elements Class 11 Important Questions Long Answer Type
Question 1.
Discuss the pattern of variation in the oxidation state of (i) B to TI and (ii) C to Pb.
Answer:

Boron and aluminium exhibit only +3 oxidation state because due to absence of d and d-electrons, they do not show inert pair effect. Elements from Ga to Tl exhibit +1 and +3 oxidation state. On moving down the group, tendency of+1 oxidation state increases because tendency of ns2 electrons of valence shell for bond formation goes on decreasing. This is known as inert pair effect. Tl+ is more stable than Tl+3.

Carbon and silicon exhibit only +4 oxidation state. Tendency to represent +2 oxidation state in heavier members increases in the order Ge < Sn < Pb. This is due to class tendency of ns2 electrons of valence shell towards bond formation (inert pair effect). Sn forms compounds in both the states and compounds of lead are more stable in +2 oxidation state than in +4 state.
Question 2.
Write diagonal relationship in Be and Al.
Answer:
Elements placed diagonally in second and third period show resemblance in properties. This is known as diagonal relationship.
1. Both possess same electronegativity.
Be =1.5 Al = 1.5 .
2. Polarizing power of Be+2 and Al+3 is nearly same.
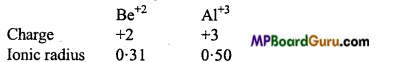
3. Alkaline earth metals are soft, but beryllium is hard like aluminium.
4. Like Al, Be is also passive to concentrated nitric acid.
5. Like Aluminium carbide, Be2C reacts with water to liberate methane.
Be+2C + 2H+2O →2BeO + CH4
Al4C3 + 12H2O → 4Al(OH)3 +3CH4
6. Both Be arid Al react with NaOH and librate hydrogen.
Be + 2NaOH → Na2BeO2 + H2
2Al+ 2NaOH +2H2O →2NaAlO2 +3H2
7. Oxides of both are amphoteric.
BeO + 2 HCl →BeCl2 +H2O
BeO+2NaOH → Na2BeO2 +H2O
Al2O3 + 6HCl → 2AlCl3 + 3H2O
Al2O3 +2NaOH →2NaAlO2 + H2O
8. BeCl2 and AlCl3 exist in the form of dimer and polymer.
9. Hydroxides of both are insoluble in water and dissociate on heating.
Be(OH)2 →BeO + H2O
2Al(OH)3 → Al2O3 +3H2O
10. BeCl2 and AlCl3 are strong Lewis acids.
11. Both the metals react with halogens to form halides.
Be + Cl2 →BeCl2
2Al + 3Cl, → 2AlCl3
12. Halides of both show covalent behaviour and are soluble in organic solvents.
Question 3.
Write similarities and dissimilarities in B and Al.
Answer:
Similarities :
- Electronic configuration of valence shell of both is ns2np.
- Covalency of both is 6.
- Oxidation number of both is +3.
- Both form M2O3 type of oxides.
- Compounds of both act as strong Lewis acids.
- Oxides of both are of amphoteric nature.
Dissimilarities:
| Boron | Aluminium |
| 1. Boron is a non-metal. | 1. Aluminium is a metal. |
| 2. Boron is a bad conductor of heat and electricity. | 2. Aluminium is a good conductor of heat and electricity. |
| 3. Its melting point is very high. | 3. Its melting point is very low. |
| 4. It does not react with dilute HCl and H2SO4. | 4. It reacts with dilute HCl and H2SO4 to liberate H2. 2Al + 3H2SO4 → Al2(SO4)3 + 3H2 |
| 5. It reacts with cone. HNO3B + 3HNO3 → H3BO3 + 3NO2 | 5. It is passive towards cone. HNO3. |
| 6. It reacts with metals to form borides. | 6. 3Mg + 2B → Mg3B2 It reacts with metals to form alloys. |
| 7. Maximum covalency of boron is 4. | 7. Maximum covalency of Al is 6. |
| 8. Its carbide is covalent. | 8. Its carbide are ionic and hydrolyse to give methane. |
Question 4.
Boron shows anomalous behaviour with other members of the group. Ex-plain.
Answer:
Boron shows anomalous behaviour with other members of the group because :
- Atomic and ionic radius of boron is less.
- Boron is a bad conductor of electricity, where as others are good conductors.
- Boron forms covalent compounds where as other members of the group form ionic compound.
- Compounds of boron are insoluble in water but soluble in organic solvents where as compounds of other members are soluble in water.
- Boron does not form trivalent ion like other members.
- Maximum covalency of boron is 4 where as maximum covalency of other members is 6.
- Oxides of boron is acidic where as oxides of other members are either amphoteric or of basic nature.
- Boron reacts with other metals to form borides where as other members react with metals to form alloys.
- Boron forms more than one type of hydride, where as other members form only one hydride.
Question 5.
What are boranes? Write their characteristics and uses.
Answer:
Hydrides of boron are known as boranes. Boron forms hydrides in two series :
Nido borane series : Its general formula is BnHn+4. Its first member BH5 does not exist. Second member B2H6 is most important which is known as diborane. Other important members are pentaborane B5H9, hexaborane B6H10.
Borane series : Its general formula is BnHn+6. Important member of this series is tetraborane B4H10, pentaborane B5H11, hexaborane B6H12.
Method of preparation:
1. Diborane is obtained by the reaction of BX3 with lithium hydride at 450K temperature.
![]()
2. Diborane is obtained by the reduction of Boron trihalide with lithium aluminium tetrahydride.
4BCl3 +3LiAlH4→ 2B2H6+ 3LiCl+3AlCl3
Characteristics :
- Diborane is a colourless gas where as other members are volatile solid.
- Diborane after combustion in presence of oxygen releases heat. Therefore, it is used as a rocket fuel.
- It is stable at low temperature. It starts dissociating at a higher temperature.
Uses :
- In the form of a rocket fuel.
- As a catalyst in polymerization reaction.
![]()
Question 6.
Show the structure of diborane. Or, Draw the structure of diborane.
Answer:
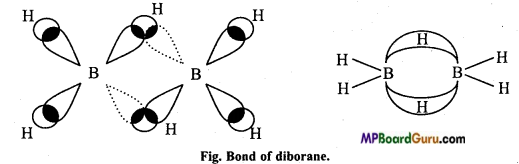
Structure of diborane: It is very interesting because it does not have sufficient valence electrons. So it is called electron-deficient compounds. Its structure is determined by electron diffraction studies.
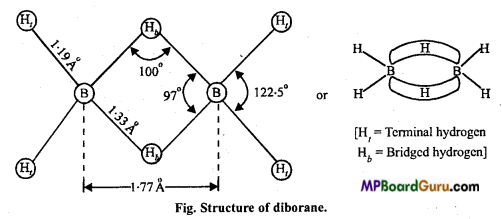
Terminal hydrogens (4) and both boron atoms are in one plane and bridged hydrogens are perpendicular to this plane in both sides. The four-terminal B-H bonds are normal electron-pair bond while B-H-B bonds are electron deficient in which 2 electrons join 3 atoms. Thus, it is called 2 electrons 3 centre or (2e – 3c) bond.
Question 7.
Compare boron and carbon.
Answer:
Similarities:
1. Both boron and carbon are non-metals.
2. Both show allotropy,
3. form more than one type of hydride.
4. Compounds of both are covalent.
5. Com- both are soluble in organic solvents.
6. Crystalline form of boron is hard like
7. Both CO2 and B2O3 dissolve in alkali and form carbonate and borate.
2NaOH +CO2 →Na2CO3 +H2O
2NaOH + B2O3 → Na2B2O3 + H2O.
Dissimilarities:
| Boron | Carbon |
| 1. Electronic configuration of boron is 1s2,2s22p1. | Electronic configuration of carbon is 1s2,2s22p2. |
| 2. Covalent of boron is 3. | Covalent of carbon is 4. |
| 3. Compounds of boron are electron deficient. | Carbon forms double or triple bond. |
| 4.Boarn does not double or triple Bond. | Compounds of boron are not electron deficient. |
| 5. Compounds of boron are Lewis acids. | Compounds of carbon are not Lewis acids. |
Question 8.
Write diagonal relationship between boron and silicon.
Answer:
Elements placed diagonally in second and third period show similarity in properties. This is known as diagonal relationship.
Diagonal relationship :
1. Both boron and silicon does not exist in free state in nature. They are found as compounds.
2. Both have same density and electronegativity.
![]()
3. Both are non-metals and exhibit allotropy.
4. Both have high melting point.
5. Both are bad conductor of electricity.
6. Both does not form cation.
7. Both do not react with dilute HCl.
8. Oxides of both react with base to form borate and silicate.
B2O3 +6NaOH → 2Na3BO3+ 3H2O
SiO2 + 2NaOH → Na2SiO3 + H2O
9. Both form various covalent hydrides.
10. Both form halide with halogen which get hydrolysed with water.
4BF3 +2H2O →HBO2 +3HBF4
3SiF4+3H2O → H2SiO3+2H2SiF6
11. Both react with nitrogen at high temperature to form nitride.
2B + N2 →2BN
2Si + 4N2 →2SiN4
12. Both the elements react with metals at high temperature to form boride and silicate.
3Mg +2B → Mg3B2
2Mg + Si → Mg2Si.
Question 9.
Compare the structure of BCl3 and AlCl3.
Answer:
BCl3 is an electron-deficient compound which always exist as a monomer. In BCl3, B is in sp2 hybridized state. Therefore, its structure is trigonal and bond angle is 120°. Because atomic radius of boron is small and chlorine bridge is unstable, therefore it does not form dimer structure.
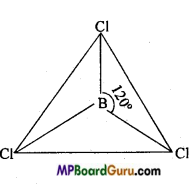
AlCl3 always exist as a dimer. In this dimer structure, each Al atom accept lone electron pair from the chlorine atom linked to other Al and complete its octet and achieve stability.
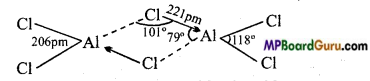
Question 10.
Explain the structure of diborane and boric acid.
Answer:
Structure of diborane: In diborane structure, four-terminal hydrogen and two boron atoms are in one plane. There are two bridged hydrogen atoms above and below this plane. The 4 terminal B – H bonds are normal bonds where as two bridged (B – H – B) are of different types, they are known as banana bond (or three centred bond).
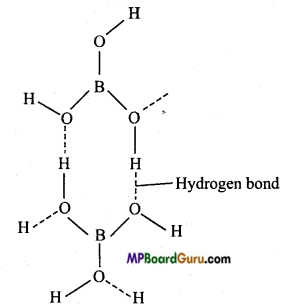
Structure of boric acid: Boric acid has layered structure, in which H3BO3 units are joined by hydrogen atoms.
Question 11.
Explain Goldschmidt aluminothermic process with a labelled diagram. Or, Explain the thermite welding process.
Answer:
Thermite process: It is also called Goldschmidt aluminothermic process. In this process, refractory crucible is filled with oxide of the metal to be reduced, aluminium powder and barium peroxide. After filling magnesium ribbon it is fixed in sand. Now, the magnesium ribbon is set on fire. A very high temperature is produced at which metal oxide get reduced by aluminium and collected in molten state. Reaction is extremely exothermic and the O2 required is supplied by barium peroxide. Selection of reducing metal is done on the basis of nature of ore.
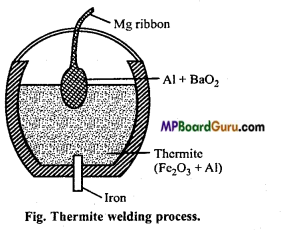
Cr2O3+2Al → Al2O3 +2Cr + Heat
Fe2O3+2Al →2Fe + Al2O3
Question 12.
What is Alum? Write its general formula, method of preparation, properties and uses. Or, Explain the method of preparation of common alum. Write any four uses of it.
Answer:
Alums: Double salts which can be represented by the general formula R2SO4. M2 (SO4)3. 24H2O are called Alums. Here R=A monovalent metal Na, K, Rb, Cs or NH4 radical
M = Trivalent metal like Fe, Al or Cr.
Alums in which trivalent metal is Al, are named as the alums of monovalent metal or radical present in them like :
Potash alum K2SO4. Al2(SO4)3.24H2O
Ammonium alum (NH4)2SO4. Al2(SO4)3.24H2O.
Methods of preparation: On crystallizing a mixture of equimolecular proportion of potassium sulphate and aluminium sulphate solution.
K2SO4 + Al2 (SO4)3 + 24H2O →K2SO4. Al2 (SO4)3.24H2O
Properties: 1. Colourless, octahedral crystals are formed whose aqueous solution is acidic due to hydrolysis. Solid alum is soluble in water but insoluble in alcohol. It’s one molecule contain 24 molecules of crystallized water.
2. On heating it melt at 92°C, on heating up to 200°C all the water of crystallization is lost and alum swells up and becomes porous. This type of alum is known as burnt alum.
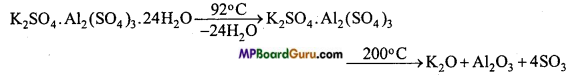
Uses :
- To stop the flow of blood and in medicines. Blood contains negative charge and due to positive charge of Al3+ coagulation takes place which stops bleeding.
- In dyeing and printing cloth, in sizing of paper.
- In purifying water.
- In special foam extinguishers.
Question 13.
What happens when :
(a) Borax is heated strongly,
(b) Boric acid is added to water,
(c) Aluminium is treated with dilute NaOH,
(d) BF3 is reacted with ammonia.
Answer:
(a) When borax is heated strongly, sodium metaborate and glass-like transparent beads of boric anhydride are obtained.
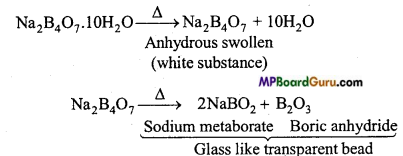
Sodium metaborate Boric anhydride Glass like transparent bead
(b) Boric acid is partially soluble in cold water where as readily soluble in hot water. It acts as a weak monobasic acid. It is not a protic acid but by accepting a hydroxide ion from water and donating a proton it behaves like a Lewis acid.
H-OH + B(OH)3 →[B(OH)4]–+H+
(c) When aluminium is treated with dilute NaOH, dihydrogen is released.
2Al(s) + 2NaOH(aq) + 6H2O(l) → 2Na+[ Al(OH)4]–+ 3H2(g)
(d) BF3, being a Lewis acid accepts an electron pair from NH3 and forms a complex compound.
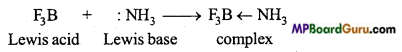
Question 14.
Carbon shows anomalous behaviour in comparison to other members of its group. Why?
Answer:
Carbon shows anomalous behaviour in comparison to other members of the group because:
1. Atomic and ionic radius is less.
2. Ionisation energy is high.
3. High electron affinity.
4. Absence of d-orbital.
Anomalous behaviour:
- Melting and boiling points of carbon is higher than other members.
- Tendency of catenation of carbon is higher than the other members.
- Carbon forms multiple bonds where as other members do not form multiple bonds.
- Monoxide of carbon is known where as that of other members are not known.
- Maximum covalency of carbon is 4 where as maximum covalency of other members is 6.
- Carbon like other members, does not form complex compounds.
- Carbon forms more than one type of hydride where as other members form only one type of hydride.
- CCl4 does not hydrolyse where as tetrahalides of other members easily hydrolyse.
- CO2 is a gas where as dioxide of other members are solid.
Question 15.
Explain the following reactions :
(a) Silicon is heated with methyl chloride at high temperature in the presence of copper.
(b) Silicon dioxide is treated with hydrogen fluoride.
(c) CO is heated with ZnO.
(d) Hydrated alumina is treated with aqueous NaOH solution.
Answer:
(a) On heating silicon with methyl chloride at high temperature in the presence of copper mixture of mono, di, tri methyl chlorosilicon and tetramethylsilane is formed.
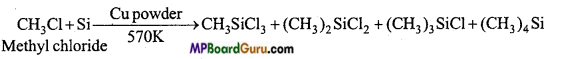
(b) On treating silicon dioxide with hydrogen fluoride, silicon tetrafluoride is obtained which dissolves in HF to form hydro fluorosilicic acid.
SiO2 +4HF → SiF4 +2H2O
SiF4 + 2HF → H2SiF6
(c) CO is a strong reducing substance but it does not reduce ZnO because ΔG° for CO → CO2, is always higher than corresponding value of ZnO. Thus, no reaction takes place.
(d) Alumina dissolves in NaOH to form sodium metaluminate.
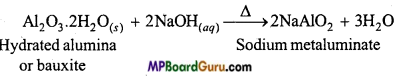
![]()
Question 16.
Write notes on the following :
1. Freon,
2. Silicones.
Answer:
1. Freon: Dichloro difluoro methane are called freons, freons are formed by the reaction of carbon tetrachloride with HF or SF3 in presence of SbCl5. Freon is used as a refrigerant in refrigerators and A.C.
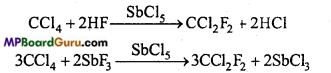
2. Silicones: Silicone is a synthetic polymer whose basic unit is R2SiCl2. Its molecular formula is like ketone, therefore these are named silicones. By the reaction of alkyl halide and Si in the presence of Cu at 575 K temperature dialkyl dihalide silane is obtained.
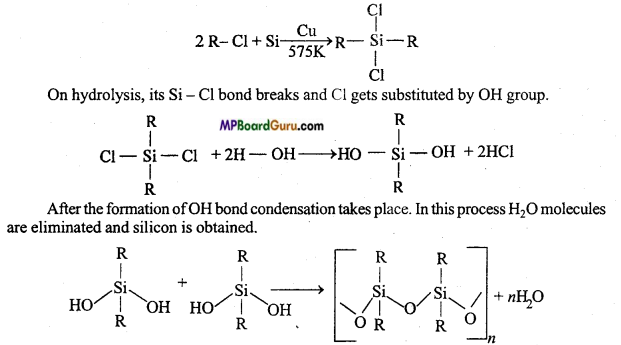
Properties :
1. Chemically inert,
2. Water repellant,
3. Insulators,
4. Unaffected by heat or heat resistant.
Use :
1. As waterproof paper,
2. As a lubricant.
Question 17.
Give reasons :
(i) Cone. HNO3 can be transported in aluminium container.
(ii) A mixture of dilute NaOH and aluminium pieces is used to open drain.
(iii) Graphite is used as lubricant.
(iv) Diamond is used as an abrasive.
(v) Aluminium alloys are used to make aircraft body.
(vi) Water should not be kept in aluminium utensils overnight.
(vii) Aluminium wire is used to make transmission cables.
Answer:
(i) Al reacts with cone. HNO3 and develops a protective layer of aluminium oxide on its surface which prevent its further reactions.

Thus, Al becomes inactive. That is why cone. HNO3 is transported in aluminium container.
(ii) NaOH reacts with A1 to liberate dihydrogen gas. This hydrogen gas is used to open drains.
2Al(s) + 2NaOH(aq)+2H2O(l) → 2NaAlO2(aq) + 3H2(g)
(iii) Graphite has layered structure. These layers are joined mutually by weak van der Waals’ attractive forces. Thus, can easily slide over one another. That is why graphite is used as a dry lubricant.
(iv) In diamond, each sp3 hybridized atom is linked with four other carbon atoms by which three dimensional lattice is formed. It is difficult to break this large covalent bond. Thus, diamond is the hardest substance found on earth. That is why it is used as an abrasive.
(v) Alloy of aluminium like duration is light, strong and corrosion-resistant. That is why, it is used in making aircraft body.
(vi) Aluminium reacts with water and oxygen (present in water) and forms a thin layer of poisonous aluminium oxide only on the walls. Therefore, water should not be kept in aluminium utensils overnight.
2Al(s) + O2(g) + H2O(l) → Al2O3(s) + H2(g)
(vii) Aluminium has high electrical conductivity. It is used for the preparation of trans¬mission cables. Again on the basis of weight its electrical conductivity is double than Cu.
Question 18.
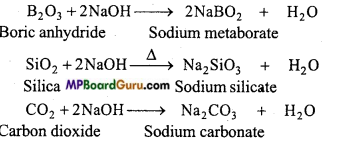
(a) Classify the following oxides into neutral, basic, acidic and amphoteric oxides:
CO, B2O3, SiO2, Al2O3, PbO3, PbO2, Tl2O3.
(b) Write the chemical reactions to show their nature.
Answer:
(ii) Due to amphoteric nature Al2O3 and PbO2, react both with acids and alkalies.
Al2O3 + 3H2SO4 → Al2 (SO4)3 + 3H2O
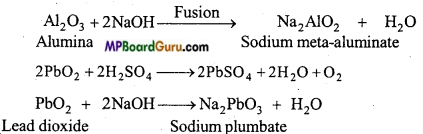
(iii) Due to basic nature TlO3 reacts with acids.
Tl2O3 + 6HCl → 2TlCl3 + 3H2O
Question 19.
Write a brief note on fullerenes.
Answer:
Fullerenes is a crystalline allotrope of carbon, but due to its ball like structure and presence of 60 carbon atom in its spherical crystal, such type of crystals are like dust particles. Formula of one unit of crystal is C60, C70, C80. C60 fullerene is also known as Buckminster fullerene. On evaporating graphite in an electric arc in helium or argon medium and condensing it, dust like powder is collected.

Properties: Fullerenes are like dust particles which are soft and spherical. Dissolves in organic solvents to give a coloured solution. Reacts with alkali metal like sodium to give Na3C60 compound. Fullerenes polymerize in presence of ultraviolet rays though their crystals do not break at 1375K temperature.
Structure: In fullerenes, there are 20, six carbon atom rings, 12 five carbon atom rings. All five atomic rings are joined to six atomic rings to which a spherically symmetrical shape is obtained. Therefore, C60 fullerene is also known as Buckyball. It is a cage-like football, radius of each sphere is 700pm.
Uses :
1. As a lubricant,
2. Compounds formed with alkali metals as semiconductors.
Question 20.
What do you understand by the following :
(a) Inert pair effect,
(b) Allotropy,
(c) Catenation.
Answer:
(a) Inert pair effect: The property of 5-electrons not to participate in bond formation is called inert pair effect. This is because the energy required to unpair the ns2 electrons is more than the energy released in the formation of the two bonds. Havier elements of group – 13, 14, 15 exhibit lower oxidation state than the number of electrons present in their valence shell. For example, in T1 +1 oxidation state is more stable than +3 oxidation state.
(b) Allotropy: The existence of an element in two or more forms which possess different physical properties but similar chemical properties, then these forms are known as allotropes and the phenomenon is known as allotropy. This is either due to difference in the number of atoms in the molecule (like O2 and O3) or due to difference in the arrangement of atoms in the molecule [like graphite, diamond and fullerene (crystalline allotrope of carbon)].
(c) Catenation: The property of self linking of atoms of the same type to form long, open or closed chain is called catenation. This property is maximum in carbon and goes on decreasing on moving down the group. In group-14, the order is as follows :
C >> Si >> Ge = Sn >> Pb
Question 21.
A certain salt X gives the following result:
(i) Its aqueous solution is alkaline to litmus.
(ii) It swells up to a glass material Y on strong heating.
(iii) When cone. H2SO4 is added to a hot solution of X, white crystal of an acid Z separates out.
Answer:
(i) Aqueous solution of salt is alkaline which represents that X is a strong base and a weak acid.
(ii) On strong heating, salt X swells up to a glassy solid Y. This represents that salt X is borax.
(iii) Hot aqueous solution of borax, reacts with cone.H2SO4 to form crystals of orthoboric acid.
Equations involved in the above reactions are as follows :
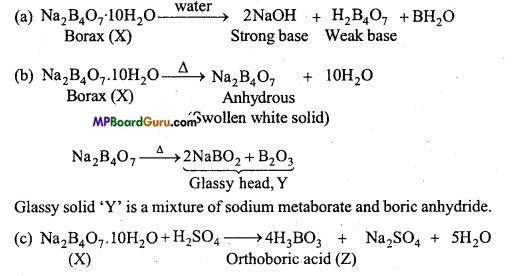
Question 22.
Write method of preparation, properties and use of carbon monoxide.
Answer:
Preparation of CO :
1. Carbon monoxide is obtained by the incomplete combustion of carbon or hydrocarbon.
2. CO is obtained by the dehydration of formic acid with cone. H2SO4.
![]()
Physical properties :
- Density of this gas is 1.25 gm/litre.
- It is a colourless, odourless, poisonous gas.
- Its boiling point is 81 K.
- Partially soluble in water.
- Strong reducing agent.
Chemical properties :
1. Combustibility: Carbon monoxide undergoes combustion in the presence of air to form C02.
2CO + O2 → 2CO2
2. Reducing property: Carbon monoxide is a strong reducing agent. It reduces metal oxides to metals.
Fe2O3 + 3CO → 2Fe + 3CO2
CuO + CO → Cu + CO2
3. It reacts with chlorine and forms a poisonous gas named phosgene.
CO + Cl2 → COCl2
4. Manufacture of metal carbonyl: Due to the presence of non-bonded electron pair on carbon monoxide, it acts as a ligand and manufactures metal carbonyls by forming a co-ordinate bond with transition metals.
Ni + 4CO →Ni (CO)4
Fe + 5CO → Fe (CO)5
Uses :
- In industries carbon monoxide is used as a fuel (as water gas and producer gas).
- As a reducing agent.
- In industrial preparation of methanol and formic acid.
- In the purification of nickel.
![]()
Question 23.
Write a note on zeolite.
Answer:
Zeolite is a type of complex silicate in which few silicon atoms are replaced by Al+3 ion. To balance the difference in valency of Si+4 and Al+3 ion some other ions like Na+, K+, Ca+2, Mg+2 etc. are present which keeps the molecule electrically neutral. Their general formula is Mx[(AlO2)x(SiO2)y ].mH2O.
Example: Na2[Al2Si3O10].2H2O
Ca[Al2Si7O18].6H2O
Zeolites have a honeycomb-like structure which has pores and channels of various size. Size of these pores is between 260 pm to 740 pm. Molecules or ions of appropriate size can be absorbed in these pores and by the channels liberation or absorption of molecules like water can take places. Therefore, these are also known as molecular sieves and they act as selecting size catalysts. Reactions catalyzed by them depends on the size of pores present in them and also on the size of reactants and products.
Structure: 24 units of tetrahedral SiO-44 ions join to form one block of zeolite. This dense octahedral block is known as octahedral block or sodalite cage. These blocks of sodalite join mutually by four-membered ring to form a two dimensional or three-dimensional network. Due to this type of structure zeolites are extremely porous. If blocks of sodalite cage are joined by double six-membered ring then the network formed is known as fouge sight.
Question 24.
What are allotropes? Sketch the structure of two allotropes of carbon namely diamond and graphite. What is the impact of structure on the physical properties of two allotropes?
Answer:
When an element exists in two or more than two forms which show different physical properties but same chemical properties then these forms are called allotropes and the phenomenon is known as allotropy.
Crystalline carbon mainly exist in two allotropes
(i) Graphite and
(ii) Diamond. In 1985 a third allotrope fullerene of carbon was discovered by H. W. Croto, E. Smale and R. F. Kurl.
In diamond each carbon atom is sp3 hybridized and linked to four other carbon atoms in a tetrahedral geometry. In diamond a three dimensional lattice of carbon atoms is formed. In graphite each carbon is sp2 hybridized and forms three sigma bonds with three nearest carbon atoms. It has layered structure and these layers are joined by weak van der Waals’ force of attraction.
Structure of two allotropes diamond and graphite of carbon and effect on their physical properties :
Diamond is a tetrahedral molecule, forming three- dimensional network. Each carbon has sp3 hybridization and is linked with four other carbon atoms by strong covalent bonds as shown in the Fig. It is clear from the figure that each carbon is situated at the centre of a regular tetrahedron and other four carbon atoms are present at the four corners of it. Experimental data proves this structure. In this structure, C-C bond length is 154 A (154 picometer).
Hence, in structure of diamond there is three-dimensional network of strong covalent bonds. It is for this reason that the diamond is the hardest substance known and it has a very high melting point (m.p. of diamond is 3843 K). All the valencies of carbon take part in the formation of C – C bonds and therefore, diamond is a bad conductor of electricity.

Structure: In graphite, each carbon is sp2 hybridized. Each carbon is linked with three other carbon atoms by single covalent bonds. The fourth electron of each carbon remains free and this accounts for good conductivity of graphite. The C – C bond length in graphite is 1.42Å (142 picometer). All the three sp2 hybridized orbitals lie in the same plane (are planar). Hence, in graphite carbon atoms form a hexagonal ring and lie in the same plane.
These rings combine together to form a plane. In graphite, there are many such planes which lie one over the other at a distance of 3.4Å. Weak van der Waals’ forces are acting in between these planes. Due to this reason, it is possible to slip one plane of graphite over other and this is responsible for lubricating property of graphite and its soft nature.

(i) Due to its hardness, diamond is used as an abrasive and in making dyes where as being soft in nature, graphite is used as pencils and as a dry lubricant in machines.
(ii) Diamond is not a conductor of electricity where as graphite is a good conductor of electricity because in it an electron of each carbon atom remains in free state.
(iii) Diamond is transparent where as graphite is not transparent.
Question 25.
Write Lewis structure and resonance structure of CO2.
Answer:
Structure: Lewis structure of CO2 is as follows :
![]()
Standard heat of CO2 is ΔH0f = -393.5kJ /mol-1 and C – O bond length is 115 pm. By this, it is clear that stability of CO2 is even more than the stability that should be obtained on the basis of Lewis structure of CO2.
This is only possible when resonance structure of CO2 is possible. Carbon dioxide is a resonance hybrid of the following resonating structures.
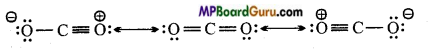
Orbital structure: In CO2, carbon is in sp hybridized state and both the oxygen are in sp2 hybridized state. In each oxygen in two sp2 orbitals non-bonding electron pair are present. One sp2 orbital overlaps with sp orbital of carbon to form σ-bond. pz orbital of one oxygen undergoes lateral overlapping with the pz orbital of carbon to form π-bond. py orbital of other oxygen laterally overlaps with p orbital of carbon to form π-bond.
This way, CO2 molecule is linear and its dipole moment is zero.

Question 26.
1. What is the basic difference in the silicones formed by the hydrolysis of R2SiCl2 and RSiCl3.
(2) [SiF6]-2 is known whereas [SiCl6]-2 is not. Why?
Answer:
1. By the hydrolysis of R2SiCl2, linear polymer of silicones is formed.
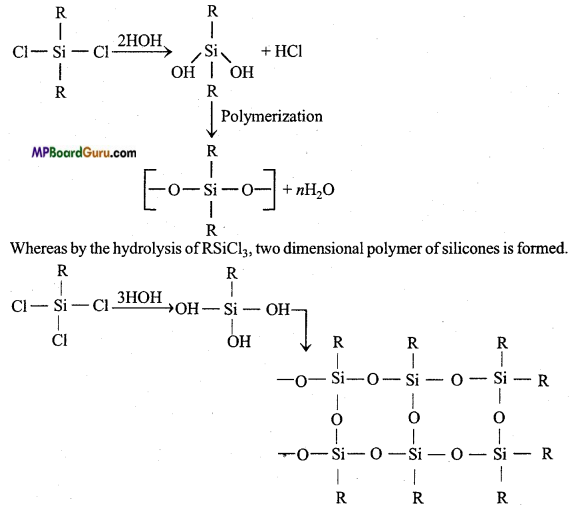
2. Size of fluoride ion is small, therefore silicon atom can combine with 6 fluoride ions. Therefore [SiF6]-2 is known where as due to larger size of chloride, silicon cannot combine with six chloride ions therefore [SiCl6]-2 is not known.
Question 27.
Give one method for industrial preparation and one for laboratory preparation of CO and CO2 each.
Ans. (a) Carbon monoxide :
(i) Industrial preparation: On passing steam over hot coke, CO is obtained.
![]()
(ii) Laboratory method: CO is obtained by the dehydration of formic acid in the presence of cone. H2SO4.

Formic acid (b) Carbon dioxide :
(i) Industrial preparation: CO2 is obtained by heating limestone.
![]()
(ii) Laboratory method: CO2 is obtained by the action of dilute HCl on CaC03.
CaCO3(s) +2HCl(aq) →CaCl2(aq) +CO2(g)+H2O(l)
Question 28.
Write short notes on :
(i) Sodium zeolite,
(ii) Sodium silicate,
(iii) Silicones.
Answer:
(i) Sodium zeolite: Sodium zeolite is also known as sodium permutit. Permutit is mixed silicate of sodium and aluminium. Its formula is : Na2[Al3Si2O8.xH2O]
Sodium zeolite is used in converting hard water into soft water. Water containing calcium and magnesium ion is known as hard water. When hard water is passed through permutit kept in a column then calcium and magnesium ions are displaced by sodium. So-dium salts do not make water hard and this way soft water is obtained. If sodium permutit is represented by Na2P then the following reaction can be written for softening of water.
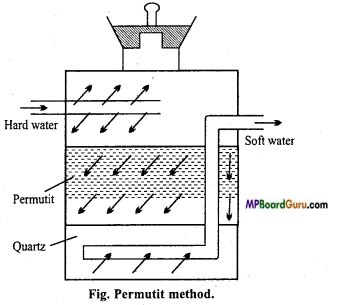
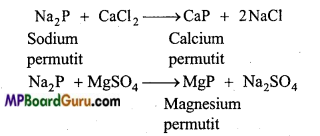
(ii) Sodium silicate or water glass: It is bright like glass and soluble in water, there-fore it is known as water glass. It contains sodium metasilicate and silica in excess and its formula is Na2SiO3 SiO2 orNa2SiO3.3SiO2.
It is obtained by fusing a mixture of sodium carbonate and sand.
Na2CO3+2SiO2 → Na2SiO3. SiO2 + CO2
The product obtained is hard, solid like glass and is soluble in water.
If sand, crystals of copper sulphate, ferrous sulphate, nickel sulphate, cadmium nitrate, manganese sulphate and cobalt nitrate etc. are added to a saturated solution of sodium silicate in a tube, then after two or three days coloured plants seem to grow in the solution and this is known as silica garden.
(iii) Silicones: These are synthetic resins of silicon and organic compounds. They are generally prepared from sand, sodium chloride and petroleum. It can be obtained in the form of gas, sticky liquid, solid like rubber or hard like stone. These are organosilicon polymers.
![]()
Question 29.
What is zeolite? Explain with example. Write any four uses of it.
Answer:
Zeolite:
Zeolite is a type of complex silicate in which few silicon atoms are replaced by Al+3 ion. To balance the difference in valency of Si+4 and Al+3 ion some other ions like Na+, K+, Ca+2, Mg+2 etc. are present which keeps the molecule electrically neutral. Their general formula is Mx[(AlO2)x(SiO2)y ].mH2O.
Example: Na2[Al2Si3O10].2H2O
Ca[Al2Si7O18].6H2O
The p-Block Elements Class 11 Important Questions Objective Type
1. Choose the correct answer:
Question 1.
Which statement is false for boric acid :
(a) It is mono basic acid
(b) It is formed by hydrolysis of boron halide
(c) Its shape is planar
(d) It is tribasic acid.
Answer:
(a) It is mono basic acid
Question 2.
Aqueous solution of Borax is :
(a) Basic
(b) Acidic
(c) Neutral
(d) Amphoteric.
Answer:
(a) Basic
Question 3.
Hybridization of boron atom in diborane is :
(a) sp
(b )sp2
(c) sp3
(d ) sp3d2.
Answer:
(b )sp2
Question 4.
Cause of polymerization of boric acid is :
(a) Acidic nature
(b) H-bond
(c) Mono basic acid
(d) Its geometry.
Answer:
(b) H-bond
Question 5.
Main ore of aluminium is :
(a) Bauxite
(b) Dolomite
(c) Galena
(d) Felspar.
Answer:
(a) Bauxite
Question 6.
Three centred-two electron bond is present in :
(a) NH3
(b) B2H6
(c) BCl3
(d) Al2Cl3.
Answer:
(b) B2H6
Question 7.
Carborundum is:
(a) B4C
(b) SiC
(c) Al3C4
(d) CaC2.
Answer:
(b) SiC
![]()
Question 8.
Which halide is electron deficient:
(a) CCl4
(b) NCl3
(c) Cl2O
(d) BCl3.
Answer:
(d) BCl3.
Question 9.
Stable allotrope of carbon is :
(a) Diamond
(b) Graphite
(c) Coal
(d) Anthracite.
Answer:
(b) Graphite
Question 10.
Best conductor of heat is :
(a) Cu
(b) B
(c) Ag
(d) Diamond.
Answer:
(b) B
Question 11.
Which does not possess electrical conductivity :
(a) K
(b) Graphite
(c) Diamond
(d) Na.
Answer:
(c) Diamond
Question 12.
Amphoteric oxide is:
(a) CaO
(b) CO2
(c) SiO2
(d) SnO2.
Answer:
(c) SiO2
Question 13.
Hardest known substance is :
(a) Coke
(b) Carborundum
(c) Corundum
(d) Diamond.
Answer:
(d) Diamond.
2. Fill in the blanks:
1. When alumina contain both Fe2O3 and SiO2 as impurity it is concentrated by ………………… process.
Answer:
Hall’s
2. When bauxite contain excess of silica as impurity, then it is concentrated by ………………… process. In this process ………………… is mixed and is heatd to 1800°C in a current of nitrogen.
Answer:
Serpeck, coke
3. Bauxite ore containing silica as impurity is mixed with coke and heated to 1800°C in a current of nitrogen when ………………… is formed and volatile silicon is separated.
Answer:
Aluminium nitride (AIN)
4. Hydrolysis of AIN formed by Serpeck process form ………………… which is heated to obtain Al2O3.
Answer:
Neutral
5. Aqueous solution of some oxides of group 13 elements convert blue litmus to red and red litmus to blue. Solution of this type of oxides are called ………………… .
Answer:
Al2Cl6
6. Aluminium chloride exist in the form of a dimmer. Its chemical formula is ………………… .
Answer:
Al(OH)3
7. Boron halide forms electron ………………… compound.
Answer:
Deficient
8. Solid CO2 is known as ………………… .
Answer:
Dry ice
9. Main factor of depletion of ozone layer is ………………… .
Answer:
Chlorofluoro carbon
10. Carbon monoxide reacts with chlorine in the presence of sunlight to form a poison-ous gas ………………… .
Answer:
Phosgene
11. First scientist to form artificial diamond is ………………… .
Answer:
Moissan
12. Germanium is a ………………… .
Answer:
Metalloid
13. Graphite is a ………………… and diamond is a of electricity.
Answer:
Good conductor,’bad conductor
14. Silicon carbide is known as …………………. .
Answer:
Carborundum.
![]()
3. Match the following:
I.
| ‘A’ | ‘B’ |
| 1. B2O3 | (a) Amphoteric |
| 2. TiO2 | (b) Acidic |
| 3. Al2O3 | (c) Basic |
| 4. Ca2O3 | (d) Basic |
| 5. Ln2O3 | (e) Amphoteric. |
Answer:
1. (c) Basic
2. (c) Basic
3. (d) Basic
4. (a) Amphoteric
5. (e) Amphoteric.
II.
| ‘A’ | ‘B’ |
| 1. In the preparation of artificial silk (cellulose xanthate) | (a) CCl4 |
| 2. In cutting and drilling diamond | (b) C2H2 |
| 3. In used for extinguishing fire in fire extinguishes | (c) B4C |
| 4. Is used in welding as oxyacetylene flame | (d) Graphite |
| 5. Black lead is known as Plumbago | (e) CS2. |
Answer:
1. (e) CS2
2. (c) B4C
3. (a) CCl4
4. (b) C2H2
5. (d) Graphite.
4. Answer in one word/sentence:
1. +1 oxidation state of T1 is more stable as compared to +3.
Answer:
Inert pair effect
2. What are hydrides of Boron known as?
Answer:
Borane
3. Which is known as two electron-three centre bond.
Answer:
Diborane
4. What will happen on mixing excess of ammonia solution in copper sulphate solution?
Answer:
A complex compound cupric ammonium sulphate is formed
5. Write the formula of alum.
Answer:
K2SO4. Al2(SO4)3.24H2O
6. What is the effect of heat on borax?
Answer:
Transparent bead of sodium metaborate + boric anhydride is formed
7. What is the name of C6O carbon crystal?
Answer:
Buckminster Fullerene
8. Which carbide is harder than diamond?
Answer:
Boron carbide
9. What is dry ice?
Answer:
Solid carbon dioxide
10. Name the compound which harm ozone layer.
Answer:
C.F.C. or freon
11. Due to which property of carbon, its number of compounds is large?
Answer:
Property of catenation
12. Name the good conductor allotrope of carbon.
Answer:
Graphite
13. What is water glass?
Answer:
Sodium silicate.