MP Board Class 9th Science Solutions Chapter 2 Is Matter Around Us Pure
Is Matter Around Us Pure Intext Questions
Is Matter Around Us Pure Intext Questions Page No. 15
Question 1.
What is meant by a pure substance?
Answer:
It is the substance which is made up of only one kind of matter. It cannot be separated into other kinds of matter by any physical process.
Examples:
- Water
- Sugar.
Question 2.
List the points of differences between homogeneous and heterogeneous mixtures.
Answer:
| Homogeneous mixture | Heterogeneous mixture |
| 1. Uniform composition throughout the mass. 2. It has no visible boundaries of separation between the constituents. 3. Constituents are not visible by naked eyes. 4. Example: Water + salt and water + sugar. | 1. Non – uniform composition throughout the mass. 2. It has visible boundaries of separation between the various constituents. 3. Constituents are visible by naked eyes. 4. Example: Powder + stones. |
Is Matter Around Us Pure Intext Questions Page No. 18
Question 1.
Differentiate between homogeneous and heterogeneous mixtures with examples.
Answer:
Homogeneous mixtures:
- These mixtures have uniform composition throughout their mass in which substances are completely mixed with one another.
- They show no visible boundaries of separation between the constituents.
- They do not show tyndall effect.
- Examples: Sugar solution, salt solution.
Heterogeneous mixtures:
- These mixtures have non – uniform composition throughout their mass and its substances remain separated and do not completely mix with one another.
- They show visible boundaries of separation between the constituents.
- They show tyndall effect.
- Examples: Sugar – soil mixture, water – oil mixture.
![]()
Question 2.
How are sol, solution and suspension different from each other?
Answer:
Sol:
- They appear to be homogeneous but actually they are heterogeneous.
- The size of solute particles is more than true solution (1 nm) but less than suspension (100 nm).
- Particles cannot be separated by Alteration.
- Particles are not visible to naked eyes.
- They scatter a beam of light.
- Examples: Smoke, blood and ink.
Solution:
- They are homogeneous mixtures.
- The size of solute particles is less than 1 nm in diameter.
- Particles cannot be separated by filteration.
- Particles are not visible to naked eyes.
- They do not scatter a beam of light.
- Examples: Sugar in water and salt in water.
Suspension:
- They are heterogeneous mixtures.
- The size of solute particles is more than 100 nm in diameter.
- Particles can be separated by filteration.
- Particles are visible to naked eyes.
- They scatter a beam of light.
- Examples: Dust in air, mud in water.
Question 3.
To make a saturated solution, 36 g of sodium chloride is dissolved in 100g of water at 293K. Find its concentration at this temperature.
Answer:
We know,
![]()
Now, mass of solute sodium chloride = 36g
Mass of Solvent (water) = 100g
So, Mass of Solution = mass of solute + mass of solvent
⇒ (36 + 100)g = 136g
Then,
Concentration of solution = 3600 / 136 × 100
⇒ 26.47% by mass.
Is Matter Around Us Pure Intext Questions Page No. 24
Question 1.
How will you separate a mixture containing kerosene and petrol (difference in their boiling points is more than 25°C) which are miscible with each other?
Answer:
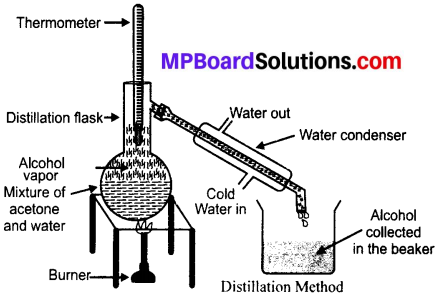
The difference in the boiling points of kerosene and water is 25°C. So, they can be separated by the process of simple distillation.
Method:
- Take the mixture in a distillation flask and fit a thermometer in it.
- Make the arrangement as shown.
- Heat slowly and observe.
- Petrol has lower boiling point, so it vaporises first and get condensed in the condenser and finally get collected in the receiver.
- The kerosene has higher boiling point, so it will be left behind.
![]()
Question 2.
Name the technique to separate:
- Butter from curd.
- Salt from sea – water.
- Camphor from salt.
Answer:
- Centrifugation
- Evaporation
- Sublimation.
Question 3.
What type of mixtures are separated by the technique of crystallisation?
Answer:
Crystallisation is used to separate impurities present in the solvent from mixtures to obtain pure substance as a crystal.
Example:
- Obtaining salt from sea – water
- purification of copper sulphate.
Is Matter Around Us Pure Intext Questions Page No. 24
Question 1.
Classify the following as chemical or physical changes:
(i) Cutting of trees.
(ii) Melting of butter in a pan.
(iii) Rusting of almirah.
(iv) Boiling of water to form steam.
(v) Passing of electric current through water and the water breaking down into hydrogen and oxygen gases.
(vi) Dissolving common salt in water.
(vii) Making a fruit salad with raw fruits and
(viii) Burning of paper and wood.
Answer:
Chemical changes: (iii), (v), (viii)
Physical changes: (i), (ii), (iv), (vi), (vii)
Question 2.
Try segregating the things around you as pure substances or mixtures.
Answer:
- Pure substances: Salt, sugar, water, silver, diamond, alcohol.
- Mixtures: Milk, air, plastic, cold – drink, bronze.
Is Matter Around Us Pure NCERT Textbook Exercises
Question 1.
Which separation techniques will you apply for the separation of the following?
- Sodium chloride from its solution in water.
- Ammonium chloride from a mixture containing sodium chloride and ammonium chloride.
- Small pieces of metal in the engine oil of a car.
- Different pigments from an extract of flower petals.
- Butter from curd.
- Oil from water.
- Tea leaves from tea.
- Iron pins from sand.
- Wheat grains from husk.
- Fine mud particles suspended in water.
Answer:
- Evaporation
- Sublimation
- Alteration
- Chromatography
- Centrifugation
- Using separating funnel
- Filteration
- Magnetic separation
- Winnowing
- Decantation and filtration.
![]()
Question 2.
Write the steps you would use for making tea.
Use the words: solution, solvent, solute, dissolve, soluble, insoluble, filterate and residue.
Answer:
Steps:
- Take 2 – 3 cups of water in a container as a solvent and boil it for few minutes.
- Add 3 – 4 teaspoons of sugar, 2 teaspoons of tea-leaves as a solute making it a solution and boil it.
- Add 1 cup of milk as a solute which will make a dark brown colour mixture. Leave it to boil.
- Sugar will get dissolved in solution but tea-leaves remain insoluble. So, it will not get dissolved in the mixture.
- Boil the mixture for few more minutes.
- Filter the solution through tea-strainer and collect the filterate in a cup.
- The tea – leaves being insoluble will be left as residue.
Question 3.
Pragya tested the solubility of three different substances at different temperatures and collected the data as given below (results are given in the following table as grams of substances dissolved in 100 grams of water to form a saturated solution).
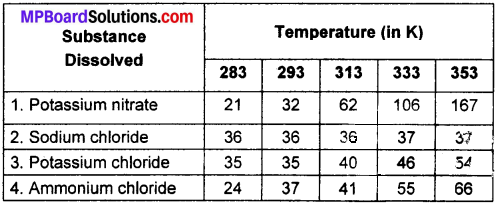
(a) What mass of potassium nitrate would be needed to produce a saturated solution of potassium nitrate in 50 grams of water at 313K?
(b) Pragya makes a saturated solution of potassium chloride in water at 353K and leaves the solution to cool at room temperature. What would she observe as the solution cools? Explain.
(c) Find the solubility of each salt at 293K. Which salt has the highest solubility at this temperature?
(d) What is the effect of change of temperature on the solubility of a salt?
Answer:
From the table,
(a) Mass of KnO3 to make saturated solution, at 313 K in 100g water at 313K = 62g
So, In 50 g of water mass of KnO3 required = (62 / 2)g = 31g.
(b) The solubility of potassium chloride (KCl) decreases on cooling and crystal formation will start.
(c) Solubility of each salt at 293 K is:
- Potassium nitrate : 32g / 100g of water
- Sodium chloride : 36g / 100g of water
- Potassium chloride : 35g / 100g of water
- Ammonium chloride : 37g / 100g of water
So, Ammonium chloride has highest solubility at 293K.
(d) On increasing the temperature, the solubility increases and on decreasing the temperature, the solubility decreases.
Question 4.
Explain the following giving examples:
(a) Saturated solution
(b) Pure substance
(c) Colloid
(d) Suspension
Answer:
(a) Saturated solution: The solution in which no more solute can be dissolved at a particular temperature is called saturated solution.
Example:
- In aqueous sugar solution, if no more sugar can be dissolved at that temperature, then it will be called as saturated solution.
(b) Pure substance: It is the substance which contains only one kind of particle and its composition is same throughout.
Examples:
- Water
- sugar.
(c) Colloid: It is the heterogeneous mixture in which the size of particles lie between true solutions and suspensions. The particles can not be seen through naked eyes but they can scatter a beam of light and possess Tyndall effect.
Examples:
- Smoke
- milk
- blood.
(d) Suspension: It is a heterogeneous mixture in which solute particles remain undissolved and get suspended within the medium. The particles scatter a beam of light and are visible through naked eyes.
Examples:
- Mud in water
- sand in water.
Question 5.
Classify each of the following as a homogeneous or heterogeneous mixtures soda water, wood, air, soil, vinegar, filtered tea.
Answer:
- Homogeneous mixture: Soda water, air, vinegar, filtered tea.
- Heterogeneous mixture: Wood, soil.
![]()
Question 6.
How would you confirm that a colourless liquid given to you is pure water?
Answer:
Boil the water at 100°C. If it boils at 100°C and left no residue behind, then it is pure water.
Question 7.
Which of the following materials fall in the category of a “pure substance”?
(a) Ice
(b) Milk
(c) Iron
(d) Hydrochloric acid
(e) Calcium oxide
(f) Mercury
(g) Brick
(h) Wood
(i) Air.
Answer:
Pure substances are
(a) Ice
(c) Iron
(e) Calcium oxide
(f) Mercury.
Question 8.
Identify the solutions among the following mixtures.
(a) Soil
(b) Sea water
(c) Air
(d) Coal
(e) Soda water.
Answer:
Solutions are:
(b) Sea water
(e) Soda water
(c) Air.
Question 9.
Which of the following will show “Tyndall effect”?
(a) Salt solution
(b) Milk
(c) Copper sulphate solution
(d) Starch solution.
Answer:
(b) Milk
(d) Starch solution.
Question 10.
Classify the following into elements, compounds and mixtures:
(a) Sodium
(b) Soil
(c) Sugar solution
(d) Silver
(e) Calcium carbonate
(f) Tin
(g) Silicon
(h) Coal
(i) Air
(j) Soap
(k) Methane
(l) Carbon dioxide
(m) Blood.
Answer:
Elements:
(a) Sodium
(d) Silver
(f) Tin
(g) Silicon.
Compounds:
(e) Calcium carbonate
(k) Methane
(l) Carbon dioxide
(j) Soap.
Mixtures:
(c) Sugar solution
(b) Soil
(h) Coal
(i) Air
(m) Blood.
Question 11.
Which of the following are chemical changes?
(a) Growth of a plant.
(b) Rusting of iron.
(c) Mixing of iron filings and sand.
(d) Cooking of food.
(e) Digestion of food.
(f) Freezing of water.
(g) Burning of a candle.
Answer:
(a) Growth of a plant.
(b) Rusting of iron.
(d) Cooking of food.
(e) Digestion of food.
(g) Burning of a candle.
Is Matter Around Us Pure Additional Questions
Is Matter Around Us Pure Multiple Choice Questions
Question 1.
Which of the following is a pure substance?
(a) Paint
(b) Sodium chloride
(c) Soil
(d) Milk.
Answer:
(b) Sodium chloride
Question 2.
Which of the following is not a mixture?
(a) Air
(b) Milk
(c) Kerosene
(d) Petroleum.
Answer:
(c) Kerosene
![]()
Question 3.
A mixture is made up of __________ .
(a) Two or more compounds.
(b) Two or more elements.
(c) Two or more elements or compounds.
(d) None of these.
Answer:
(c) Two or more elements or compounds.
Question 4.
Husk in water is an example of __________ .
(a) Colloid
(b) Solution
(c) Suspension
(d) Saturated solution.
Answer:
(c) Suspension
Question 5.
Which of the following is a metalloid?
(a) Iron
(b) Boron
(c) Zinc
(d) Carbon.
Answer:
(b) Boron
Question 6.
A colloidal solution is also called:
(a) True solution
(b) Sol
(c) Solvent
(d) Real solution.
Answer:
(b) Sol
Question 7.
When a ray of light passes through a solution and particles are not visible, then it is called:
(a) True solution
(b) Suspension
(c) Colloid
(d) Heterogeneous solution.
Answer:
(a) True solution
Question 8.
Which of the following is a true solution?
(a) Muddy water
(b) Salt solution
(c) Blood
(d) Curd.
Answer:
(b) Salt solution
Question 9.
The zig – zag motion of colloidal particles is known as __________ .
(a) Brownian movement
(b) Tyndall effect
(c) Rectilinear movement
(d) Electrolysis.
Answer:
(a) Brownian movement
Question 10.
A separating funnel is used for separating __________ .
(a) Solids
(b) Miscible liquids
(c) Immiscible liquids
(d) None of these.
Answer:
(c) Immiscible liquids
Question 11.
Particles of mixture of ammonium chloride and sand can be separated by __________ .
(a) Sublimation
(b) Evaporation
(c) Centrifugation
(d) Decantation.
Answer:
(a) Sublimation
![]()
Question 12.
A solution that can be separated by filtration is called __________ .
(a) Colloid
(b) Suspension
(c) True solution
(d) None of these.
Answer:
(b) Suspension
Question 13.
The separation technique which involves the difference in their densities is __________ .
(a) Centrifugation
(b) Sublimation
(c) Distillation
(d) Filteration.
Answer:
(a) Centrifugation
Question 14.
Milk of Magnesia is a:
(a) True solution
(b) Colloid
(c) Suspension
(d) Homogeneous mixture.
Answer:
(c) Suspension
Question 15.
Concentration of solution is __________ .
(a) mass of solute / mass of solvent × 100
(b) mass of solvent / mass of solute × 100
(c) mass of solute / mass of solution × 100
(d) mass of solution / mass of solute × 100.
Answer:
(c) mass of solute / mass of solution × 100
Question 16.
Which one of the following is not a chemical change?
(a) Freezing of water
(b) Ripening of fruit
(c) Formation of curd
(d) Corrosion of metals.
Answer:
(a) Freezing of water
Question 17.
Tincture of iodine is the solution of __________ .
(a) Iodine in water
(b) Iodine in acid
(c) Iodine in alcohol
(d) Iodine in bromine.
Answer:
(c) Iodine in alcohol
Question 18.
An alloy is a __________ .
(a) Heterogeneous mixture
(b) Homogeneous mixture
(c) Colloid
(d) Both (a) and (b).
Answer:
(b) Homogeneous mixture
Question 19.
Tyndall effect in forest is seen due to the presence of:
(a) Air in forest
(b) Dust in water
(c) Water droplets in air
(d) All of the above.
Answer:
(c) Water droplets in air
Question 20.
The compound FeS is not attracted by magnet because __________ .
(a) It is a compound
(b) It is black
(c) Iron has lost its properties
(d) It is a liquid.
Answer:
(c) Iron has lost its properties
Is Matter Around Us Pure Very Short Answer Type Questions
Question 1.
Define pure substance.
Answer:
The substance which is made up of only one kind of particle.
Question 2.
Give four examples of pure substance.
Answer:
Iron, aluminium, water, sodium chloride.
![]()
Question 3.
Define metalloids.
Answer:
Elements which show properties of metals as well as non – metals.
Question 4.
Name the scientist who first used the term element.
Answer:
Robert Boyle (1661).
Question 5.
Name two elements which are liquid.
Answer:
Mercury and bromine.
Question 6.
Give two examples of homogeneous mixtures.
Answer:
Salt solution, copper sulphate solution, brass alloy.
Question 7.
Give examples of heterogeneous solution.
Answer:
Sugar and sand, mud in water, iron filings in powder.
Question 8.
Give examples of colloidal solution.
Answer:
Blood, ink, milk.
Question 9.
Name the types of colloids.
Answer:
Sol, solid sol, aerosol, emulsion, foam, gel, solid foam.
Question 10.
Name the solutions which show Tyndall effect.
Answer:
Muddy water, milk of magnesia, starch solution, milk and ink.
Question 11.
Name the process used in separation of cream from milk.
Answer:
Centrifugation.
Question 12.
Name the technique used in separation of oil from water.
Answer:
Separation using separating funnel.
Question 13.
What is emulsion?
Answer:
If both dispersed phase and dispersing medium is liquid, it is called emulsion. Example: Milk.
Question 14.
Calculate the concentration of solution which contains 3 g of salt dissolved in 60 g of water.
Answer:
Concentration = Mass of solute / mass of solution × 100
⇒ (3 / 60 + 3) × 100%
⇒ 300 / 63%
⇒ 4.76%.
![]()
Question 15.
What happens to solubility of solid in liquid when the temperature increases?
Answer:
Increases.
Question 16.
What are the other names of solute and solvent?
Answer:
Dispersed phase and dispersed medium.
Question 17.
Give two examples of aerosol.
Answer:
Fog, hairspray.
Question 18.
Which one has smallest particles – colloid, suspension or solution?
Answer:
Solution.
Question 19.
Name the substances that can be separated by sublimation.
Answer:
Iodine, ammonium chloride and camphor.
Question 20.
Which process is used to separate ink from coloured component (dye)?
Answer:
Evaporation.
Is Matter Around Us Pure Short Answer Type Questions
Question 1.
Define:
(a) Pure substance
(b) Mixture
(c) Metalloid
(d) Solution
(e) Solute
(f) Solvent
(g) Concentration of solution
(h) Saturated solution
(i) Unsaturated solution
(j) Emulsion
(k) Distillation
(l) Tyndall effect
(m) Crystallisation.
Answer:
(a) Pure substance: A pure substance is made up of only one kind of particle. It includes all elements or compounds.
Examples:
Iron, gold, silver, water, sugar, hydrochloric acid.
(b) Mixture: It is the impure substance which is made up of two or more different types of pure substances like elements or compounds in any ratio.
Examples:
Milk, rocks air, salt solution and soil.
(c) Metalloid: These are the elements which show the properties of metals and non – metals. They are also called semi – conductors.
Examples:
Silicon (Si), germanium (Ge) and boron (B).
(d) Solution: It is the homogeneous mixture of two or more substances.
Examples:
Air, alloys, salt solution.
(e) Solute: It is the component of solution which is dissolved in the medium.
Example:
salt in salt solution.
(f) Solvent: It is the component of solution which dissolves the solute.
Example:
water in salt solution.
(g) Concentration of solution: It is the quantity of solute present in the given quantity of solution.
(h) Saturated solution: It is the solution in which no more solute can be dissolved at that particular temperature.
(i) Unsaturated solution: It is the solution is which more amount of solute can be dissolved at the temperature to make it saturated.
(j) Emulsion: It is a colloid in which a liquid is dispersed in another liquid but they are immiscible.
Examples:
Milk, beauty cream.
(k) Distillation: It is the technique which is used to separate two miscible liquids which are having different boiling points.
Examples:
Separating pure water from sea – water, separation of mixture of acetone and water.
(l) Tyndall effect: It is the effect caused due to scattering of light by colloidal particles.
Example:
Sunlight when passes through forest get scattered due to tiny water droplets present in the air.
(m) Crystallisation: It is the process of obtaining pure solid substances in the form of crystals from its saturated solution by cooling it down slowly.
![]()
Question 2.
Give two examples of each:
- Element
- mixture
- homogeneous mixture
- heterogeneous mixture
- solution
- suspension
- colloid.
Answer:
- Element: Iron, gold.
- Mixture: Soil with powder, sand in water.
- Homogeneous mixture: Sugar solution, salt solution.
- Heterogeneous mixture: Soil in water, milk.
- Solution: Sugar solution, salt solution.
- Suspension: Milk of magnesia, chalk with water.
- Colloid: Ink, milk.
Question 3.
Write all types of colloidal solution and give examples.
Answer:
Sol : Ink, paints
Aerosol : Fog, spray paint
Emulsion : Butter
Foam : Fire – extinguisher foam, shaving foam
Gel : Hair gel
Solid sol : Colourful gemstones.
Question 4.
Write down the properties of a true solution.
Answer:
- It is a homogeneous mixture.
- The particles are not visible to naked eyes even not with a microscope.
- Particles passes through filter paper.
- True solution does not show Tyndall effect.
Question 5.
Write down the properties of a suspension.
Answer:
- It is a heterogeneous mixture.
- The particles are visible to naked eyes.
- Particles do not pass through a filter paper.
- It shows Tyndall effect.
Question 6.
Define physical and chemical change and give two examples of each.
Answer:
(i) Physical change: It is a change in which no new substance is formed and particles do not change their characteristics.
Examples:
- Formation of ice from water
- cutting of paper.
(ii) Chemical change: It is a change in which new substance is formed and particle changes their characteristics.
Examples:
- Ripening of fruits
- burning of wood.
Question 7.
Define chromatography. Give examples where it is used.
Answer:
Chromatography: It is the method of separation of solid solutes which are dissolved in the same solvent. A special paper called “Chromatography paper” is used in this technique.
Examples:
- Separation of dyes present in the ink
- Separation of pigments from natural colours.
Question 8.
Name the separation technique used in the separation of:
(a) Iron pins from soil.
(b) Butter from curd.
(c) Copper sulphate from impure sample.
(d) Pigments from extract of flower.
(e) Different gases from air.
(f) Mixture of salt – water.
(g) Mixture of salt and ammonium chloride.
(h) Mixture of immiscible liquid – petrol and water.
Answer:
(a) Magnetic separation.
(b) Centrifugation.
(c) Crytallisation.
(d) Chromatography.
(e) Fractional distillation.
(f) Distillation.
(g) Sublimation.
(h) Separating funnel.
Question 9.
What is Brownian movement?
Answer:
The particles of colloids move randomly in a zig – zag manner. This movement is called Brownian movement. It happens due to collision of particles of dispersed phase and dispersion medium.
Question 10.
Write the dispersed phase and dispersion medium of the following colloidal substances: Mist, milk, paint, sponge, coloured gemstone, ink, smoke, face cream.
Answer:
| Colloidal Substance | Dispersed Phase | Dispersion Medium |
| Mist | Gas | Liquid |
| Milk | Liquid | Liquid |
| Paint | Liquid | Liquid |
| Sponge | Solid | Gas |
| Coloured gemstone | Solid | Solid |
| Ink | Liquid | Liquid |
| Smoke | Gas | Solid |
| Face cream | Liquid | Liquid |
Question 11.
21.6g of sodium chloride dissolves in 60g of water at 20°C. Calculate its solubility.
Answer:
Now,
60g of water dissolves = 21.6 g of sodium chloride
So, 100g of water dissolves = (21.6 / 60) × 100g of sodium chloride
⇒ 36g of sodium chloride
Thus, solubility of sodium chloride is 36g at 20°C.
Question 12.
As solution contains 60 ml of alcohol mixed with 200 ml of water. Calculate the concentration of this solution.
Answer:
Concentration of solution = (volume of solute / volume of solution) × 100
= (60 / 200) × 100%
= 30% (by volume)
![]()
Question 13.
What are the advantages and disadvantages of crystallisation method over evaporation method in separation of mixture?
Answer:
1. Advantages:
- Proper crystals of substance are obtained.
- Soluble impurities can be removed easily.
2. Disadvantages:
- Some substance get remain with the solvent.
- Solvent needs to be purified again for further use.
Is Matter Around Us Pure Long Answer Type Questions
Question 1.
Explain the Centrifugation process? Mention its applications also.
Answer:
Centrifugation process: It is the process of separation of suspended particles of a substance from a liquid by churning the liquid at high speed. In this process, denser particles are forced to settle at the bottom and lighter particles stay at the top. The following figure shows the process of centrifugation: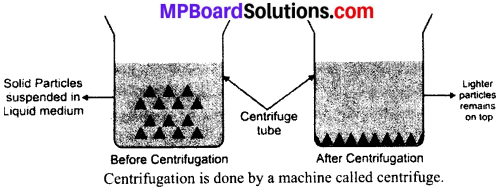
Applications:
- In washing machine, for washing clothes.
- In diagnostic labs, to test blood.
- In dairies, to separate butter from milk.
Question 2.
How can we separate the mixture of two immiscible liquids – kerosene oil and water? Explain the method with a labelled diagram.
Answer:
Mixture of two immiscible liquids – kerosene oil and water, is separated by separating funnel method. This method is based on the principle of difference in their densities.
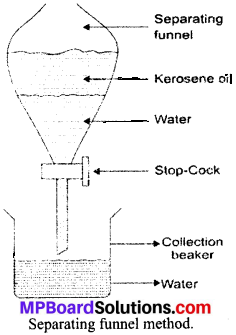
Method:
- Take the mixture of kerosene oil and water in a separating funnel as shown in the figure.
- Leave the medium undisturbed for sometime till separate layers of oil and water are formed.
- Open the stop – cock of the funnel and collect the water (denser liquid) in the collection beaker.
- Close the cock when the kerosene oil reaches the stop – cock.
- Collect the oil in a separate beaker.
Question 3.
How can mixture of salt and ammonium chloride be separated? Name the technique and the principle involved. Mention one application of this technique.
Answer:
Mixture of salt and ammonium chloride can be separated by the process of sublimation (as shown in figure). The principle of sublime nature is involved in this.
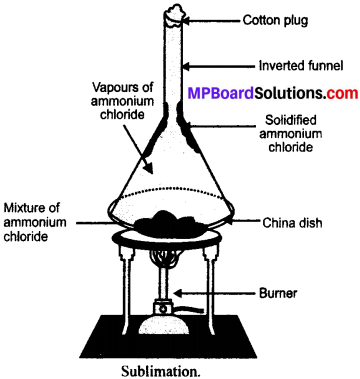
Method:
- The mixture is kept in a china dish placed on a stands shown in figure.
- An inverted funnel is kept over the mixture with cotton plug to close it.
- Place the burner below it and heat the mixture.
- On heating, ammonium chloride directly changes from solid to gaseous state and gets collected at the stem of the funnel.
- The common salt is left behind.
Application:
- Separation of naphthalene balls’ powder and Common salt.
![]()
Question 4.
Elaborate the technique to separate dyes in black ink using chromatography ?
Answer:
Chromatography is the method of separation of solid solutes which are dissolved in the same solvent.
Method of separation of dyes in black ink:
- Take a thin and long strip filter paper or chromatography paper.
- Make a line on it at 3 cm from the lower end.
- Put a small drop of black ink on it at the centre of line.
- Dry the paper.
- Dip the paper from lower edge into a tall, glass jar containing same water in a way that the ink drop remains just above the water and leave it undisturbed.
- The water start rising in the paper by capillary action and the ink drop starts splitting into the constituents.
- The constituent which is more soluble in water will rise more than less soluble constituent
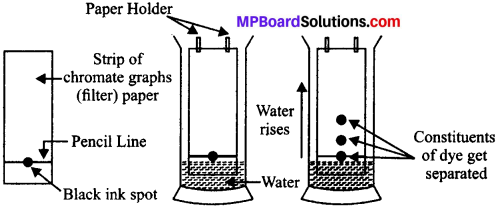
Chromatography technique is used in separation of:
- Drugs from blood.
- Pigments from natural colours.
- Sugar from urine.
Question 5.
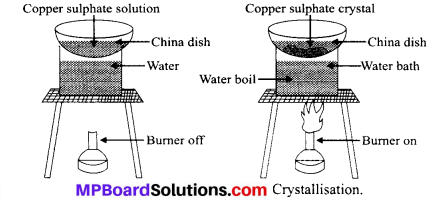
What is crystallisation? How can we obtain pure copper sulphate from impure copper sulphate solution?
Answer: Crystallisation is the process of obtaining pure solid substance in the form of crystals from its saturated solution by cooling down slowly.
Method to obtain pure copper sulphate from its impure solution:
- Take some quantity of impure copper sulphate in water in a china dish.
- Filter the solution to remove insoluble impurities.
- Take a beaker Riled half with water and place the china dish over it.
- Heat the beaker to heat the copper sulphate solution through water bath.
- The copper sulphate solution gets evaporated till the solution becomes saturated.
- Cool down the saturated solution slowly to obtain the crystals of pure copper sulphate.
- Separate the copper sulphate crystals from solution by Alteration and dry them.
Question 6.
What is distillation technique? How can the mixture of acetone and water be separated?
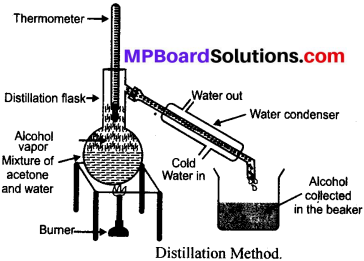
Answer:
Distillation: It is the technique used to separate two miscible liquids by conversion of liquid into vapours by heating and then condensing the vapours into the pure liquid.
Types:
(a) Simple distillation: This technique is used to separate only two liquids where difference of boiling points is more than (20 – 25°C).
(b) Fractional distillation: This technique is used to separate more than tw o liquids and also where difference of boiling points of liquids is less than (20 – 25°C).
Procedure to separate the mixture of acetone and water:
- Take the mixture of acetone and water in a distillation flask.
- Fix a thermometer to the flask.
- Heat the mixture slowly and observe the temperature of thermometer.
- Acetone vaporises first and get condensed in the condenser and finally get collected in the beaker.
- Water is left behind in the flask.
Question 7.
What is Magnetic separation method? Write the procedure to separate iron filings from sulphur powder. Write some applications.
Answer:
Magnetic separation method is used to separate magnetic component of a mixture using strong magnets like horse-shoe magnet or electro magnets.
Separation of iron filing from sulphur powder:
- Take the mixture of iron filings and sulphur powder in a china-dish.
- Take a horse shoe magnet and move it over the mixture.
- The iron filings get attached to the magnet leaving the sulphur powder in the china dish.
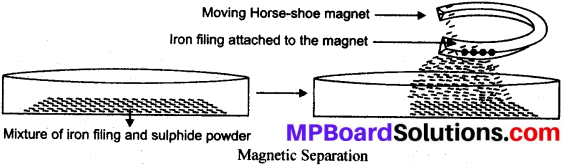
Applications:
- Separation of iron materials from waste.
- Removing iron materials from metal scrap.
- Removing of iron foreign particles from eyes using electro-magnets by the doctors.
Is Matter Around Us Pure Higher Order Thinking Skills (HOTS)
Question 1.
With the help of diagram explain the water purification system in water works.
Answer:
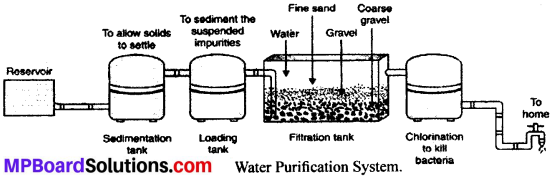
Procedure:
- Firstly, the water is collected in the reservoir.
- Then it is passed to sedimentation tank to allow solid impurities to get settle down.
- Then, it is passed to loading tank where the suspended impurities are loaded to settle down as sediment.
- Then, water is filtered into the filtration tank where it is passed through different layers of sand and gravel.
- Finally, it is passed through chlorinated tank where it is mixed with chlorine to kill bacteria or germs present in it. Now, it is called disinfected water.
- This disinfected water is pumped to high storage tank from where it is supplied to city through network of pipes.
Question 2.
Name all the separation techniques used to separate different mixtures indicating the type of mixtures as well.
Answer:
A. Separation of solid – solid mixture:
- Solvent method: Sugar and sand.
- Magnetic method: Iron filings from sulphur powder.
- Sublimation: Common salt and ammonium chloride.
B. Separation of solid – liquid mixture:
- Filtration method: Chalk and water, sand and water.
- Centrifugation method: Clay particles from water, cream from milk.
- Crystallisation method: Pure copper sulphate from impure solution.
- Chromatography: Dyes from the black ink, pigments in natural colours, sugar from blood.
- Distillation: Acetone and water, gaseous components of air.
C. Separation of liquid – liquid mixture:
- Separating funnel method: Petrol and water, chloroform and water.
Is Matter Around Us Pure Value Based Questions
Question 1.
Kritika was asked to bring the jar of common salt from kitchen. But accidentally jar got slipped into the tub of water. She got tensed of scolding by mother. She went to his brother mentioning the problem. Her brother advised her to place the tub in open sunlight for long hours for two – three days. She did that and observed that common salt is left behind.
(a) What is the technique used above to separate the common salt from water?
(b) Name the type of mixtures that can be separated from this technique.
(c) Mention the values of Kritika’s brother mentioned here.
Answer:
(a) Evaporation technique is used here to separate the common salt from water.
(b) Mixtures in which solid substance is dissolved in water can be separated by this technique.
Example:
Coloured component (dye) from ink.
(c) Krikita’s brother showed his intelligent and helpful behaviour.
![]()
Question 2.
Vaibhav and Hritik’s teacher asked them to separate the mixture of acetone and water contained in the jar. Vaibhav brought a beaker and Hritik brought a separating funnel to separate the mixture. After few minutes, Hritik come with water and acetone contained in separate containers but Vaibhav come with the same mixture. Teacher congratulate Hritik for separating the mixture and advised Vaibhav to use the same technique followed by Hritik.
(a) Which technique is followed by Hritik for separation?
(b) Name the principle involved in this technique.
(c) Mention the value of Hritik seen here.
Answer:
(a) Separating funnel method is followed by Hritik for separation.
(b) Principle of difference of densities is applied in this technique.
(c) Values of obedience and wiseness is seen by Hritik.
Question 3.
Three friends Sanjana, Saumya and Shruti were sitting in a room. Sanjana told Shruti and Saumya about the visibility of dust particles in air. But, they were doubting her statement Then Sanjana closed the room and opened a window slightly. She told diem to observe the path of light and other things in that path. Saumya and Shruti were very surprised to see the presence of minute dust particles in air.
(a) Which effect is illustrated by Sanjana?
(b) Where this phenomenon can be seen naturally?
(c) What values did Sanjana show to her friends?
Answer:
(a) Tyndall effect is illustrated by Sanjana.
(b) This phenomenon is seen naturally in dense forest, in winter season through fog.
(c) Sanjana showed the values of wiseness and friendliness.
Question 4.
Pranjal went to her friend’s house and bring a black ink and a special kind of paper with her. Her friend asked her about that paper. Pranjal perform an activity with the paper by applying the dot of ink on it. And, placed the paper in ajar of water in such a way that the dot just touched the upper level of water. Finally, they observed the separation of different coloured constituents of that ink.
(a) Name the technique used by Pranjal.
(b) Describe the technique mentioned and name the paper used.
(c) Write some other applications of this technique.
(d) Write the values shown by Pranjal.
Answer:
(a) Chromatography technique is used here.
(b) Chromatography is the method of separation of solid solutes which are dissolved in the same solvent. Chromatographic paper is used in this technique.
(c) This technique is used in separation of drugs from blood, pigments from natural colours and separating sugar from urine.
(d) Pranjal showed the values of intelligence and friendliness.