MP Board Class 9th Science Solutions Chapter 1 Matter in Our Surroundings
Matter in Our Surroundings Intext Questions
Matter in Our Surroundings Intext Questions Page No. 3
Question 1.
Which of the following are matter? Chair air, love, smell, hate, almonds, thought, cold, cold – drink smell of perfume.
Answer:
Chair, air, almonds and cold – drink.
Question 2.
Give reasons for the following observation: The smell of hot sizzling food reaches you several metres away, but to get the smell from cold food you have to go close.
Answer:
The particles of hot food have more kinetic energy due to higher temperature, so their rate of diffusion is more and they move several meters away as compared to the particles of cold food.
Question 3.
A diver is able to cut through water in a swimming pool. Which property of matter does this observation show?
Answer:
This shows that the particles of matter have space between them and a weak force of attraction between them.
![]()
Question 4.
What are the characteristics of the particles of matter?
Answer:
Following are the characteristics of the particles of matter:
- Particles of matter are very small.
- Particles of matter have space between them.
- Particles of matter are continuously moving.
- Particles of matter attract each other.
Matter in Our Surroundings Intext Questions Page No. 6
Question 1.
The mass per unit volume of a substance is called density, (density = mass / volume). Arrange the following in order of increasing density: air, exhaust from chimneys, honey, water, chalk, cotton and iron.
Answer:
The order of increasing densities:
air < exhaust from chimneys < cotton < water < honey < chalk < iron.
Question 2.
(a) Tabulate the differences in the characteristics of states of matter.
(b) Comment upon the following: Rigidity, Compressibility, Fluidity, Filling a gas container, Shape, Kinetic energy and Density.
Answer:
(a)
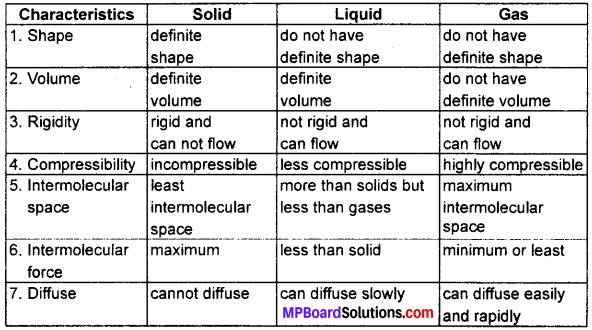
(b)
(i) Rigidity: It is the tendency of matter to retain or maintain its shape when an outside force is applied.
- Solids are rigid in matter
(ii) Compressibility: It is the property of matter due to which it can be compressed to lower volume.
- Solids have minimum compressibility, but gases have maximum.
(iii) Fluidity: It is the tendency of a matter or particles of a matter to flow.
- Liquid and gases have fluidity so they are fluids.
(iv) Filling a gas container: Gas particles fill the container completely due to negligible intermolecular force and maximum space between particles.
(v) Shape: It is the property of a matter which indicates its boundaries.
- Solid have fixed shape but liquids and gases have no fixed shape.
(vi) Kinetic energy: The energy of matter or particles of matter due to their motion is called kinetic energy.
- Particles of gases have highest kinetic energy and liquids have less kinetic energy than gases but more than solids.
(vii) Density: It is mass per unit volume.
- Solids have highest density, liquids have low density than solids but greater than gases.
![]()
Question 3.
Give reasons:
(a) A gas fills completely the vessel in which it is kept.
(b) A gas exerts pressure on the walls of the container.
(c) A wooden table should be called a solid.
(d) We can easily move our hand in air but to do the same through a solid block of wood, we need a karate expert.
Answer:
(a) The molecules of gas have very less force of attraction and possess high kinetic energy due to which they move in all directions and fill the vessel completely.
(b) The particles of gas move freely and randomly in all directions. So, they collide with each other and also with the walls of the container due to which they exert a Pressure on its walls.
(c) A wooden table has a fixed shape, fixed volume. It is rigid and cannot be compressed. So it should be called a solid.
(d) The particles of Air have very less force of attraction between them, so we can easily move our hand, but particles of wood have strong force of attraction, so we have to apply a greater amount of force to break it and pass through solid wood.
Question 4.
Liquids generally have lower density as compared to solids. But you must have observed that ice floats on water. Find out why.
Answer:
We know that ice is solid and water is liquid but the structure of ice is cage like due to which the molecules of water are not closely packed and have vacant space between them due to which ice has low density than water and floats over it.
Matter in Our Surroundings Intext Questions Page No. 9
Question 1.
Convert the following temperature to celsius scale:
(a) 300 K
(b) 573 K
Answer:
(a) 300 K = (300 – 273)°C = 27°C.
(b) 573 K = (573 – 273)°C = 300°C.
Question 2.
What is the physical state of water at:
(a) 250°C
(b) 100°C?
Answer:
(a) Gaseous state.
(b) Liquid and gaseous both.
Question 3.
For any substance, why does the temperature remain constant during the change of state?
Answer:
During the change of state of any substance, the temperature remains constant because the heat supplied to the substance is used in overcoming the force of attraction between the particles and change its state. This hidden heat is called latent heat.
![]()
Question 4.
Suggest a method to liquefy atmospheric gases.
Answer:
The atmospheric gases can be liquefied by cooling and applying pressure on them in a closed chamber or cylinder.
Matter in Our Surroundings Intext Questions Page No. 10
Question 1.
Why does a desert cooler cool better on a hot dry day?
Answer:
On a hot dry day, the rate of evaporation is high because of low humidity. So, water sprinkling on the pads of cooler gets evaporated from outside which results in making its walls cool and then we receive cool air.
Question 2.
How does the water kept in an earthen pot (matka) become cool during summer?
Answer:
The earthen pot has a lot of pores on its surface. So, water comes out and gets evaporated from these pores and cools the water inside the pot.
Question 3.
Why does our palm feel cold when we put some acetone or petrol or perfume on it?
Answer:
When we put acetone, petrol or perfume on our palm, then these liquids absorb energy from our palm and get evaporated which cause cooling effect on our palm.
Question 4.
Why are we able to sip hot tea or milk faster from a saucer rather than a cup?
Answer:
Saucer has a large surface area than a cup. So the rate of evaporation is more in saucer than cup which causes faster cooling of the hot tea. Hence, we can sip hot tea from a saucer faster than cup.
Question 5.
What type of clothes should we wear in summer?
Answer:
Light coloured cotton clothes should be worn in summer because light colour reflects heat and cotton absorbs sweat quickly and evaporates it easily which makes our body feel cool and dry.
Matter in Our Surroundings NCERT Textbook Exercises
Question 1.
Convert the following temperatures to the Celsius scale.
(a) 293 K
(b) 470 K.
Answer:
(a) 293 K into°C
⇒ 993 – 273 = 20°C
(b) 470 K into°C
⇒ 470 – 273 = 197°C
![]()
Question 2.
Convert the following temperatures to the Kelvin scale.
(a) 25°C
(b) 373°C
Answer:
(a) 25°C = 25 + 273 = 298 K
(b) 373°C = 373 + 273 = 646 K.
Question 3.
Give reason for the following observations.
(a) Naphthalene balls disappear with time without leaving any solid.
(b) We can get the smell of perfume sitting several metres away.
Answer:
(a) Naphthalene balls disappear with time without leaving any solid, because naphthalene balls sublime and directly change into vapour state without leaving any solid.
(b) We can get the smell of perfume sitting several metres away because perfume contains volatile solvent i.e., gaseous particles, which have high speed and large space between them and diffuse faster and can reach people sitting several metres away.
Question 4.
Arrange the following substances in increasing order of forces of attraction between the particles – water, sugar, oxygen.
Answer:
Oxygen → water → sugar.
Question 5.
What is the physical state of water at:
(a) 25°C
(b) 0°C
(c) 100°C?
Answer:
(a) 25°C is liquid.
(b) 0°C is solid or liquid.
(c) 100°C is liquid and gas.
Question 6.
Give two reasons to justify:
(a) water at room temperature is a liquid.
(b) an iron almirah is a solid at room temperature.
Answer:
(a) Water at room temperature is a liquid because its freezing point is 0°C and boiling point is 100°C.
(b) An iron almirah is a solid at room temperature because melting point of iron is higher than the room temperature.
![]()
Question 7.
Why is ice at 273 K more effective in cooling than water at the same temperature?
Answer:
Ice at 273 K will absorb heat energy or latent heat from the medium to overcome the heat of fusion to become water. Hence, the cooling effect of ice is more than the water at same temperature because water does not absorb this extra heat from the medium.
Question 8.
What produces more severe burns, boiling water or steam?
Answer:
Steam at 100°C will produce more severe burns as extra heat is hidden in it called latent heat. Whereas, the boiling water does not have this hidden heat.
Question 9.
Name A, B, C, D, E and F in the following diagram showing change in its state
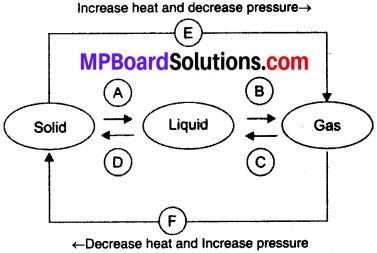
Answer:
A → Liquefication / melting / fusion
B → Vaporisation / evaporation
C → Condensation
D → Solidification
E → Sublimation
F → Sublimation
Matter in Our Surroundings Additional Questions
Matter in Our Surroundings Multiple Choice Questions
Question 1.
Which of the following substances is solid?
(a) Plasma
(b) BEC
(c) Wood
(d) Mercury
Answer:
(c) Wood
![]()
Question 2.
Fusion is the phenomenon of changing of state of __________ .
(a) Liquid to gas
(b) Solid to liquid
(c) Solid to gas
(d) Gas to plasma.
Answer:
(b) Solid to liquid
Question 3.
Diffusion is not possible in the case of __________ .
(a) Liquid into Solid
(b) Gas into liquid
(c) Gas into Gas
(d) Solid into Solid.
Answer:
(d) Solid into Solid.
Question 4.
Compressibility is highest in __________ .
(a) Liquid
(b) Solid
(c) Gas
(d) Plasma.
Answer:
(c) Gas
Question 5.
We can smell odour of deodrant several metres away due to __________ .
(a) Diffusion
(b) Evaporation
(c) Fusion
(e) None.
Answer:
(a) Diffusion
Question 6.
Which of the following do not exhibit sublimation?
(a) Water
(b) Camphor
(c) Naphthalene
(d) Dry ice.
Answer:
(a) Water
Question 7.
Ice floats on water because of __________ .
(a) Higher density than water
(b) Equal density than water
(c) Lower density than water
(d) None of these.
Answer:
(c) Lower density than water
Question 8.
As the pressure of air increases then boiling point of liquid __________ .
(a) Increases
(b) Decreases
(c) Remains same
(d) None of these.
Answer:
(a) Increases
![]()
Question 9.
The atmospheric pressure equals to __________ .
(a) 1.01325 × 108 Pa
(b) 101.3 25 × 104 Pa
(c) 1.01325 × 105 Pa
(d) 1.01325 × 102 Pa.
Answer:
(c) 1.01325 × 105 Pa
Question 10.
Cooking at high altitudes is difficult because __________ .
(a) Boiling point decreases
(b) Boiling point increases
(c) Freezing point reduced
(d) None of the above.
Answer:
(a) Boiling point decreases
Question 11.
Density of water is highest at __________ .
(a) 3°C
(b) 5°C
(c) 4°C
(d) 6°C.
Answer:
(a) 3°C
Question 12.
Dry ice is __________ .
(a) Solid carbon monoxide (CO)
(b) Solid Nitrogen dioxide (NO2)
(c) Solid carbon dioxide (CO2)
(d) Solid Ammonia (NH3).
Answer:
(c) Solid carbon dioxide (CO2)
Question 13.
The amount of heat required to change the state of 1 kg of substance is called __________ .
(a) Calorific heat
(b) Latent heat
(c) Thermal heat
(d) Conversion heat.
Answer:
(b) Latent heat
Question 14.
Evaporation takes place at __________ .
(a) Boiling point
(b) Melting point
(c) Freezing point
(d) At all temperatures.
Answer:
(d) At all temperatures.
Question 15.
Evaporation generally takes place at __________ .
(a) Below boiling point
(b) Above boiling point
(c) At boiling point
(d) None.
Answer:
(a) Below boiling point
Question 16.
Evaporation causes __________ .
(a) Heating effect
(b) Boiling effect
(c) Cooling effect
(d) Absorbing effect.
Answer:
(c) Cooling effect
Question 17.
Latent heat of vaporisation of water is __________ .
(a) 2.25 × 106 J/kg
(b) 3.34 × 106 J/kg
(c) 22.5 × 106 J/kg
(d) 33.4 × 105 J/kg.
Answer:
(a) 2.25 × 106 J/kg
Question 18.
Which factor does not affect the rate of evaporation?
(a) Humidity
(b) Colour
(c) Wind Speed
(d) Surface area.
Answer:
(b) Colour
Question 19.
The rate of evaporation is minimum in __________ .
(a) Dry day
(b) Humid day
(c) Hot day
(d) Stormy day.
Answer:
(b) Humid day
Question 20.
Which of the following processes consumes heat?
(i) Melting
(ii) Freezing
(iii) Vaparisation
(iv) Condensation.
(a) (i) and (ii)
(b) (ii) and (iii)
(c) (i) and (iii)
(d) (ii) and (iv).
Answer:
(c) (i) and (iii)
Matter in Our Surroundings Very Short Answer Type Questions
Question 1.
Define Matter.
Answer:
Matter is defined as something which occupies space and has mass.
Question 2.
What is the melting point of ice?
Answer:
0°C or 273 K.
Question 3.
Name the phenomenon by which two substances intermix with each other.
Answer:
Diffusion.
Question 4.
Name the physical State of matter which can be highly compressed.
Answer:
Gaseous state.
Question 5.
Name the tiny particles of which matter is made up of.
Answer:
Atoms.
![]()
Question 6.
Name the process of conversion of solid state into gaseous state directly.
Answer:
Sublimation.
Question 7.
Name the Indian scientist who calculated the fifth state of matter.
Answer:
Satyendra Nath Bose.
Question 8.
Name all the states of matter.
Answer:
Solid, liquid, gas, plasma, BEC (Bose Einstein Condensate).
Question 9.
What is Chemical name of dry ice?
Answer:
Solid Carbon Dioxide.
Question 10.
What is the main effect produced by evaporation?
Answer:
Cooling effect.
Matter in Our Surroundings Short Answer Type Questions
Question 1.
Define diffusion? On which features does it depend?
Answer:
It is the mixing of particles of different matters. It depends on the state, temperature and kinetic energy of particles.
Question 2.
Define:
(a) Latent heat of fusion
(b) Latent heat of vaporisation.
Answer:
(a) Latent heat of fusion is the amount of heat energy required to convert 1 kg of a solid into liquid at its melting point.
(b) Latent heat of vaporisation is the amount of heat energy required to convert 1 kg of liquid into gas at its boiling point.
Question 3.
Explain the following terms:
(a) Sublimation
(b) Evaporation.
Answer:
(a) Sublimation: The phenomenon of change of state directly from solid to gas or vapours or vice-versa without changing into liquid state.
Examples:
Dry ice, camphor.
(b) Evaporation: The process of changing matter from liquid state into the vapour state at any temperature below its boiling point.
Examples:
Drying of clothes, volatile liquids like petrol, kerosene.
Question 4.
What are the different factors on which evaporation of liquids depend?
Answer:
The factors on which evaporation depends are:
(a) Temperature
(b) Surface area
(c) Humidity
(d) Wind speed
Question 5.
Write four substances which show sublimation.
Answer:
(a) Camphor
(b) Naphthalene balls
(c) Dry ice
(d) Ammonium chloride
Question 6.
Why the temperature of ice remains constant when it starts melting?
Answer:
When the ice melts, then it absorbs the latent heat of fusion which changes its states. And, that heat is consumed in changing the state, keeping the temperature constant.
Question 7.
How can we liquefy gas? How is it useful to us?
Answer:
Gases can be liquefied by cooling them and by applying pressure into a closed chamber like cylinder. This property of compressibility is useful for us and we use it through different ways e.g., LPG (Liquefied Petroleum Gas), CNG (Compressed Natural Gas) and Liquefied O2 in welding.
Question 8.
Differentiate between:
(a) Freezing and Melting.
(b) Evaporation and Boiling.
Answer:
(a) Freezing and Melting:
| (a) Freezing | Melting |
| 1. It is the conversion from liquid state into solid state | 1. It is the conversion from solid to liquid state. |
| 2. Heat is released. | 2. Heat is absorbed. |
(b) Evaporation and Boiling:
| (b) Evaporation | Boiling |
| 1. It takes place on the surface of liquid. | 1. It happens to all liquids. |
| 2. It takes place at all temperatures. | 2. It happens at a particular temperature. |
| 3. It is slow. | 3. It is fast. |
| 4. It causes cooling. | 4. It does not cause cooling. |
Question 9.
Why solid carbon dioxide is called dry ice?
Answer:
It is because carbon dioxide is pressurised in the form of solid state which looks cool like ice. But when it is heated, it directly get converted into carbon dioxide gas.
![]()
Question 10.
Change the following temperatures accordingly.
(a) -173°C
(b) 250 K
(c) 2°C
(d) 0°K
Answer:
(a) We know that,
Temperature on kelvin = Temperature on Celsius + 273
So, -173°C = (-173 + 273) K = 100 K
(b) 250 K = (250 – 273)°C = -23°C
(c) 2°C = (2 + 273) K = 275 K
(d) 0°K = (0 – 273)°C = -273°C.
Question 11.
Give one example of each: Melting, Vaporisation, Condensation, Sublimation, Evaporation.
Answer:
Melting: Ice to water
Vaporisation: Water to steam
Condensation: LPG
Sublimation: Naphthalene balls, solid iodine
Evaporation: Volatile liquids like petrol.
Matter in Our Surroundings Long Answer Type Questions
Question 1.
Give reasons:
(а) The smell of hot tasty cooked food reaches us from far.
(b) The smell of perfume reaches us several metres away.
(c) The fragrance of an incense stick spreads in entire hall quickly.
(d) Steam is more severe than boiling water.
(e) We see water droplets collected on outer surface of steel glass containing cold water.
(f) We feel lot of perspiration on a humid day.
(g) Evaporation causes cooling.
(h) It is advised to use pressure cooker at high altitudes.
(i) We should wear light colour cotton clothes in summer.
(j) A gas fills the container completely.
(k) Clothes take more time to dry in a humid day.
(l) Kerosene oil is kept in a cool place.
Answer:
(a) The particles of hot tasty food have high kinetic energy due to raised temperature. So, they can easily diffuse and move to long distances.
(b) The particles of perfume are in vapour form. So, they possess high kinetic energy and move randomly and finally reach us several metres away.
(c) The particles of an incense stick are in gaseous state. So, they have high kinetic energy and diffuse quickly into atmosphere and cover the room.
(d) Steam has the heat in the form of latent heat as compared to boiling water. So, it is more severe.
(e) The water vapours present in the atmosphere get condensed when it comes in contact with the chilled container and get deposited in the form of liquid droplets.
(f) The rate of evaporation decreases on humid day as there are sufficient water vapours present in the atmosphere. So, our sweat does not get dried and we perspire a lot.
(g) During the evaporation, the particles on the surface of the fluid take energy and heat from the surface get vaporised which create cooling effect.
(h) It is advised to use pressure cooker at high altitudes because the atmospheric pressure is low and water boils quickly. So, to increase the pressure and to cook the food properly, pressure cooker is required.
(i) Light colour cotton clothes are good reflectors of heat as well as good absorbers of sweat. When sweating occurs, the cotton absorbs it quickly and evaporates it faster.
(j) The particles of gas have very less intermolecular force of attraction and have large intermolecular space. So, they possess high kinetic energy and expand quickly resulting in filling the container completely.
(k) The rate of evaporation is minimum on a humid day. So, cloth takes more time to get dried.
(l) Kerosene a volatile substance and it evaporates quickly and also there is a risk of breaking of bottle due to the pressure created by its particles. So, it is stored in a closed container and kept in a cool place.
Question 2.
(a) What is evaporation? On what factors does it depend?
(b) How is it useful to us?
Answer:
(a) Evaporation:
The phenomenon of changing of a matter from liquid state into the vapour state or gaseous state at any temperature below its boiling point is known as evaporation. The rate of evaporation depends on the following factors:
- Surface area: If the surface area of the liquid increases, then evaporation becomes faster.
- Temperature: When the temperature of the liquid increases then the kinetic energy of the particles increases which causes faster evaporation.
- Wind speed: When the speed of wind increases, it covers the vacant space in the atmosphere created by moving wind, the evaporation gets faster. Also, we have seen that clothes dry faster in a windy day than a normal day.
- Humidity: If the humidity is least then rate of evaporation is faster but when humidity is high, then Atmosphere can’t hold much water vapours which causes slow rate of evaporation.
(b) Uses of evaporation:
- Drying of clothes.
- Separation of different mixtures like water and salt.
- Desert coolers etc.
Question 3.
Give one example of each:
(a) Water vapours present in the atmosphere.
(b) Diffusion of a gas into liquid.
(c) Diffusion of a liquid into liquid.
(d) Diffusion of a gas into gas.
(e) Two sublime substances.
Answer:
(a) Presence of water droplets is seen on the outer surface of steel container containing cold water.
(b) Presence of dissolved oxygen in marine water which is used up by aquatic organisms.
(c) Mixing of ink and water.
(d) Burning of incense stick.
(e) Ammonium chloride, Naphthalene balls.
Question 4.
(a) What is matter? What characteristics does a matter possess?
(b) Explain all the five states of matter.
Answer:
(a) Matter: Matter is anything which occupies space and has mass.
(b) Matter has five states:
- Solid: It is the state of matter which has a fixed shape, volume, high density, incompressible and cannot flow.
- Liquid: It is the state of matter which possesses indefinite shape, definite volume, low compressibility and can flow easily.
- Gas: It is the state of matter which possesses indefinite shape, indefinite volume, high compressibility and can flow easily.
- Plasma: This state of matter consists of free electrons . and ions in the form of ionised gases.
- BEC (Bose – Einstein Condensate): This state of matter is achieved by cooling gas of extremely to low density of about one – hundredth thousand the density of a normal air to super low temperature.
Matter in Our Surroundings Higher Order Thinking Skills (HOTS)
Question 1.
Osmosis is a special kind of diffusion, Comment
Answer:
In diffusion, molecules of a substance move from higher concentration to lower concentration. But, during Osmosis, the water or solvent molecules move from their higher concentration to the place of their lower concentration. Therefore, Osmosis is a special kind of diffusion.
Question 2.
Why does honey diffuse in water at a slower rate than ink?
Answer:
Honey diffuse at a slower rate than ink since the density of honey is greater than that of water.
Matter in Our Surroundings Value Based Questions
Question 1.
Gaurav is preparing for his cricket match in summer camp. He took full sleeves black colour silk shirt for the match. But his mother advised him to wear white half sleeve cotton shirt
(a) Why did mother advise him to wear white cotton shirt?
(b) What type of clothes we should wear in summer?
(c) What values of mother are reflected here?
Answer:
(a) Mother advised him to wear white cotton shirt because white is a good reflector of heat and cotton is a good absorber of sweat and helps in quick evaporation, which will make Gaurav feel cool during the match.
(b) Light coloured cotton clothes should be worn in summer.
(c) Mother showed her values of caring and insightful.
Question 2.
Aakansha parked her scooty in a parking of her society. In the evening, she saw her scooty tyre was burst. Then, she immediately went to mechanic to repair the puncture and advised the mechanic not to inflate the tyres fully.
(а) Why did the scooty punctured?
(b) Why she advised mechanic not to inflate the tyres fully?
(c) What values of Aakansha is reflected here?
Answer:
(a) The scooty got punctured due to bursting of tyre because the particles of air expanded and gained kinetic energy due to heat and pressurised the walls of tyre resulting in bursting.
(b) She advised mechanic not to inflate the tyres fully as the summer days were hot which caused the expansion of inflated air and create excessive pressure in the tyres.
(c) Aakansha showed her values of intelligence and awareness.
![]()
Question 3.
Ansh had a “Pooja” in his house. His mother told him to bring camphor required for “Pooja” from the market. On the “Pooja” day when he opened up the packet, he got surprised to see it empty. Then, Ansh got alert and brought a new packet.
(a) Why did the packet got empty?
(b) Name the property of matter mentioned here.
(c) Why did Ansh keep new camphor packet in a closed container?
(d) What values of Ansh are mentioned here?
Answer:
(a) Packet got emptied due to the evaporation of camphor due to its sublime nature.
(b) The property of sublimation is shown here.
(c) To prevent sublimation of camphor.
(d) Ansh shows the values of curiosity, wisdom and duty.
Question 4.
Sanjana went to a farm house with her family to spend her summer vacations. But the farm house faced the frequent problem of power cut. So, the refrigerator was not chilling the water bottles. Then, she brought two earthen pots for making drinking water, cool.
(a) How did the earthern pot, cool the water in it?
(b) Name the phenomenon involved in it
(c) Mention the values of Sanjana depicted here.
Answer:
(a) Earthen pots have small pores through which water gets evaporated making the water cool inside the pot.
(b) The process of evaporation is involved in it.
(c) Sanjana showed her intelligent, caring and responsible behaviour.